What Is The Molecular Geometry Of Pbr3
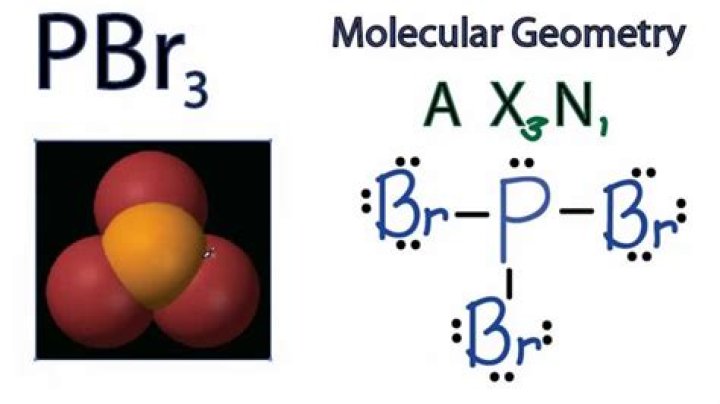
What Is The Molecular Geometry Of Pbr3
Do you use VSEPR theory to predict electron pair geometry? 3
1. Use VSEPR theory to predict electron pair geometry and molecular geometry of psrustribromide, PBr3.
a) Linear geometry of electron pairs, linear molecular geometry.
b) The electron pair is a geometry triangular planner, the geometry of a molecule is a triangular planner.
c) Electron pair geometry is triangle, molecular geometry is curved.
d) Electron pair geometry is tetrahedral, molecular geometry is tetrahedral.
e) Electron pair geometry is tetrahedral, molecular geometry is a triangular pyramid.
2. The central atom of the triiodide ion, I3, is surrounded on all sides.
a) Two single bonds and no single electron pair.
b) Two single bonds and two isolated pairs of electrons.
c) Two single bonds and three isolated pairs of electrons.
d) Single bonds, double bonds and single electron pairs.
e) Two double bonds and one electron pair.
3. Which of the following statements is true?
1. The least electrically negative atom is usually the central atom of the molecule.
2. As the difference in electrical negativity between two bonded atoms increases, the percentage of the ionic role of the bond increases.
3. In table ic each group has less electronegativity.
4. Which of the following molecules or ions is free: N2O, OCl and ClO2?
a) N2O only b) OCl only c) ClO2 only
d) N2O and ClO2 e) OCl and ClO2
To determine the geometry of a molecule, always use the Lewis structure and count the bond pairs and the single electron pairs.
Pay attention to this table:
http: // ...
Three bonds in PBr3 and 1 isolated pair == Tetrahedral electron pair geometry and triangular pyramid molecular geometry


