Br Ion Name - How To Discuss
Br Ion Name
What is the name of the acid formed by the ionic bromine Br?
The acid is bromic acid, HBrO3. Examples of bromates are sodium bromate (NaBrO3) and potassium bromate (KBrO3).
What is the name of the acid formed by the ionic bromide Br - where?
This is a bromine atom with an ionic charge of -1 (Br-), for example in cesium bromide, cesium cations (Cs +) are electrically attracted to the bromide anions (Br-) to form the electrically neutral ionic compound CsBr. Bromide.
Last name| chemical formula | friend |
| molar mass | 79. ### 904 gmol - 1 |
| conjugated acid | Hydrogen bromide |
| Thermochemistry |
friendWhat is the ionic name of bromine?
Name of the monatomic anions
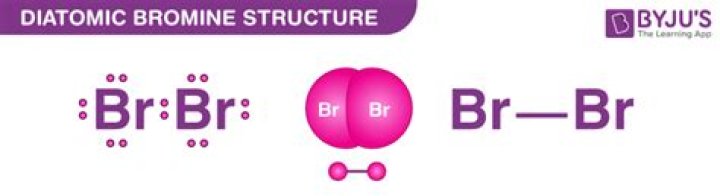
Name of the element Name of the ion Formula of the ion| bromine | bromide | friend |
| iodine | iodide | THE - |
| oxygen | oxide | O2- |
| sulfur | sulfide | S2− |
What does bromine do to the human body?
Liquid bromine is corrosive to human tissues and its vapors irritate the eyes and throat. The vapor from the source is highly toxic if inhaled. Humans can absorb organic bromine through the skin, with food and through the breath. Organic bromine is widely used as a spray to kill insects and other unwanted pests.
What is the symbol of the bromide ion?
Is HCl a cation or an anion?
HCl, which contains the chloride anion, is known as hydrochloric acid. HCN, which contains cyanide anion, is called hydrogen cyanide. Naming rules for oxyacids (the anion contains the element oxygen): Since all these acids have the same H + cation, it is not necessary to name the cation.
Can bromine kill you?
Liquid bromine is corrosive to human tissues and its vapors irritate the eyes and throat. The vapor from the source is highly toxic if inhaled. Humans can absorb organic bromine through the skin, with food and through the breath. Organic bromine is widely used as a spray to kill insects and other unwanted pests.
What is the value of the bromide?
What is the difference between bromine and bromide?
Furthermore, the main difference between bromine and bromide is that bromide is the reduced form of bromine. Bromide also has 36 electrons versus 35 electrons for bromine, but both have 35 protons. Hence, bromine has a charge while bromine is neutral.
What color is bromide?
Datasone
what is the mermaid nomenclature called?
Acids are named after their anion, the ion bonded to hydrogen. In simple binary acids, an ion is bonded to hydrogen. The names of these acids consist of the prefix hydro, the first syllable of the anions and the suffix ic. Complex acid compounds contain oxygen.
How is bromine named?
The name Brome comes from the Greek word bromos for stink, according to Los Alamos National Laboratory. The red-brown liquid easily evaporates into a red vapor with a strong chlorine-like odor. According to Lenntech, bromine is dangerous.
Is oxygen a cation or an anion?
Is bromine vital?
Bromine has proven to be vital. Bromine - an element with atomic number 35 and chemical symbol Br - is the 28th chemical element essential for tissue development in humans and all other animals, according to a team of researchers led by Professor Billy Hudson of the Vanderbilt University School of Medicines.
Where is bromine found in nature?
Bromine occurs naturally in the earth’s crust and in seawater in various chemical forms. Bromine is also found in swimming pools as an alternative to chlorine. Brominated products are used in agriculture and plumbing and as flame retardants (chemicals that prevent something from igniting).
Is phosphorus a cation or an anion?
Positive and negative ions: cations and anions
Is the CU an ion?
Copper (2+) is a copper ion that carries a double positive charge. It has a cofactor function. It is a divalent metal cation, a copper cation and a monoatomic thickening. 5.3 Related point.
Why is bromine soluble in water?
Is bromine a cation or an anion?
Magnesium is a group 2 metal that forms cations with a stable configuration of noble gas electrons when it loses both of its valence electrons. Bromine forms a stable electronic configuration of noble gas when it accepts an electron. For example, we find magnesium as a 2+ cation and bromine as anion 1 in a ratio of 1: 2 in the compound.
Is bromide an acid or a base?
acid-base approx| Last name | Last name | |
| Great | Perchloric acid | perchloration |
| 3.2 * 109 | Iodine hydrogen | iodide |
| 1.0 * 109 | Hydrobromic acid | bromide |