Po43 Lewis Structure - How To Discuss
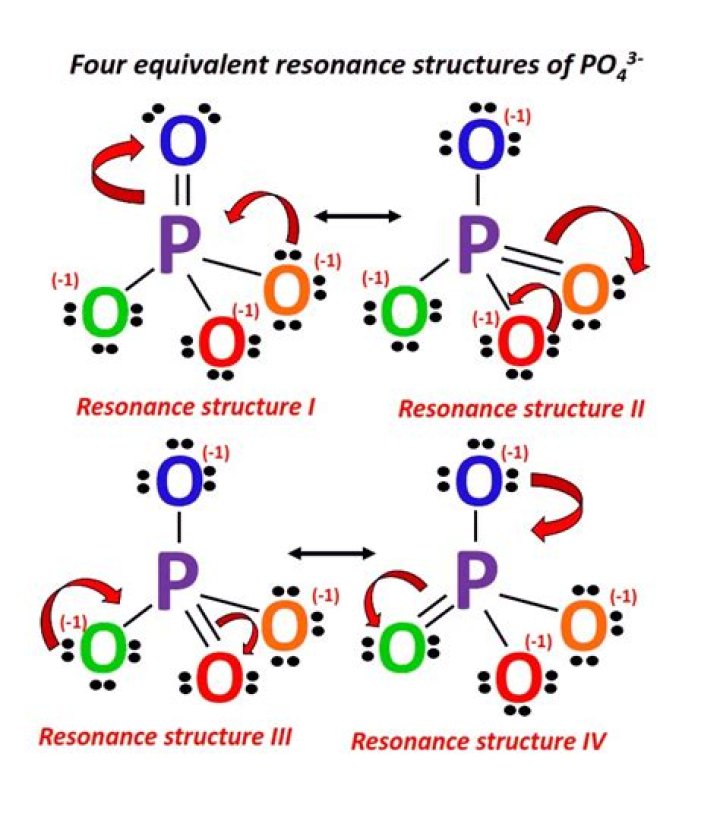
Po43 Lewis Structure
How many resonance structures does po43 make?
4 resonance structuresDoes phosphate also have resonance structures?Oxygen atoms can give phosphorus a single pair at a time, creating more resonant structures. The response to phosphating has important biochemical consequences. Any positively charged ion with three bonds to an oxygen atom is called an oxonium ion.Do you also know how many extra electrons there are in the Lewis structure for the po43 phosphate?
ThreeHow many valence electrons are there in po43?
32 valence electronsIs phosphate polar or non-polar?
The phosphate has a charge of 3, which leads to a tetrahedral arrangement of the 4 oxygen atoms around the phosphorus atom, with the distribution of electrons on the surface appearing quite homogeneous. Hence, the net dipole appears to be zero, which makes the phosphating non-polar.Is So3 a resonance structure?
There are seven resonance structures for SO3.Is po3 4 polar or non-polar?
above and below each O and which uses 32 electrons. This means we have a tetrahedral structure. So it is symmetrical, PO43 is not polar.How is a deposit order calculated?
If the molecule contains more than two atoms, follow these steps to determine the order of the bonds: Draw the Lewis structure. Count the total number of bonds. Count the number of bonding groups between each atom. Divide the number of bonds between the atoms by the total number of bonding groups in the molecule.What is the order of the titles on po43?
The bond order is equal to the total number of bonds (sigma and pi) in different resonance structures between 2 atoms in a molecule / total number of resonance structures. In the case of a bond order PO43 = 5/4 = 1.25.What does po4 mean?
Phosphorus, blood. Synonym / acronym: inorganic phosphorus, phosphate, PO4.Are po43 tetrahedra?
The tetrahedral molecular structure is observed in several molecules, the most common of which is methane, CH4. Other molecules include silane, SiH4 and thiazyl trifluoride, NSF3. The tetrahedral structure is also found in phosphating, PO43, sulfation, SO42 and perchlorination, ClO4.What is the Lewis structure of no2?
Draw the Lewis structure for NO which means it has only 7 valence electrons. In the Lewis structure of NO2, the nitrogen atom is the least electronegative atom and is at the center of the structure. For the Lewis structure of NO2, calculate the total number of valence electrons for the NO2 molecule.How much does so4 cost?
We need 32 valence electrons for 4 outer shells (8x4), but we only have 30. So the charge is 2 for the ion.How many single pairs does po4 3 have?
From the top right corner and clockwise we can create 4 resonant structures which extend the byte P to 10. In all these elements, P has a formal charge of 0 and an oxygen is also 0.total valence 32 Remaining 0 Why does phosphate have a negative charge of 3?
Phosphate is PO4. Now O 2 needs electrons to have 8 electrons in the outer shell and to become stable. So PO4 has a total of 3 more electrons than the protons it has. It is therefore negatively charged.How many resonance structures does so4 2 have?
sixwhat is the hybridization of po43?
At the top of the answer there are three PO bonds and one P = O bond ... number of electrons around P = 5 + 3 = 8 which are four pairs of electrons. Therefore. Hybridization is sp3.Po43 Lewis Structure
Po43 Lewis Structure
Prepare a Lewis structure for phosphate ion (PO43Â ')?
Create a unique Lewis structure for phosphate ions (PO43Â ') after the byte principle. Contains all formal charges and isolated pairs of electrons.
I can't just drag on this page, but ...
Keep P as the central atom.
Attach 4A atoms around it with a single binding on each atom.
Then place 3 lone pairs of electrons in each A atom.
32 available valence electrons are used.
This structure will adhere to the byte principle and its formal compensation is 1 on each of the four O atoms and +1 on the P atom (net 3 as required).
This is the correct answer to this problem.
Po43 Lewis Structure
Po43 Lewis Structure
O P (O ) (O ) = O
There are three types of oxygen associated with PK. Each of the three oxygen has a charge of 1. Fourth, oxygen is bound to PK twice, without charge.
This gives P a +5 oxidation state.
Po43 Lewis Structure
Po43 Lewis Structure
Draw the same Lewis structure for phosphate ion (PO43Â ')? ۔
Create a unique Lewis structure for the phosphate ion (PO43Â ') following the byte principle. Contains all formal charges and isolated pairs of electrons.
I can't just drag on this page, but ...
Keep P as the central atom.
Attach 4 O atoms around each atom with single bonds.
Then place 3 single pairs of electrons in each O atom.
32 available valence electrons are used.
This structure will comply with the byte principle and the formal charge is 1 on each of the four O atoms and +1 on the P atom (net 3 as required).
O P (O ) (O ) = O
There are three types of oxygen associated with PK. Each of the three oxygenes has a charge of 1.
This gives P an +5 oxidation state.
Po43 Lewis Structure
Po43 Lewis Structure
Draw a single Lewis structure for phosphate ion (PO43Â ')? 3
Create a unique Lewis structure for the phosphate ion (PO43Â ') following the byte principle. Includes all formal charges and individual electron pairs.
I can't easily drag on this page, but ...
Keep P as the central atom.
Pair 4 O atoms around each atom with a single bond.
Then place 3 lone pairs of electrons in each O atom.
32 available valence electrons are used.
This structure will adhere to the byte principle and its regular charge is 1 on each of the four O atoms and +1 on the P atom (net 3 if necessary).
Po43 Lewis Structure
Po43 Lewis Structure
O P (O ) (O ) = O
Three types of oxygen are associated with P. Each of the three oxygen charges is 1. The fourth oxygen is connected to the double uncharged P.
This gives P an oxidation state of +5.