Oxidizing agent - How To Discuss
Oxidizing agent,
Definition of Oxidizing agent:
A substance that tends to bring about oxidation by being reduced and gaining electrons.
Substance that tends to give-off oxygen when the ambient temperature rises over a certain point and, thus, may cause ignition of sensitive organic material. This term includes oxidizers such as chlorates, perchlorates, peroxides, etc., that react spontaneously and vigorously on contact with some metals and other materials. Also called oxidant. See also reducing agent.
How to use Oxidizing agent in a sentence?
- I didnt know if the substance would be the oxidizing agent in the experiment and I didnt want to find out because chemistry scared me.
- You should always have a very good oxidizing agent on hand in case you ever need one in an emergency situation.
- You should try to always have an oxidizing agent around just in case you are ever in urgent need of one.
- The oxidizing agent causes the oxidation of an atom by receiving an electron from that atom and is itself reduced.
Meaning of Oxidizing agent & Oxidizing agent Definition
What is a "good oxidizing agent" mean? Ions, atoms and molecules with strong electron affinity are considered good oxidizing agents. The stronger the electron affinity, the greater the oxidative capacity. Elemental fluorine is considered the strongest elemental oxidizing agent.
Which one is a stronger oxidizing agent?
At the same time, the best oxidants are the most elemental forms of electronegative atoms, and they note that the weakest oxidant is metallic sodium (Na+), and the strongest is the elemental fluorine molecule (F2), which can oxidize large amounts. number of substances.
Which substance behaves as the oxidizing agent?
In chemistry, an oxidizing agent (oxidizing agent, oxidizing agent) is a substance that has the ability to oxidize other substances, that is, to accept their electrons. Oxygen, hydrogen peroxide and halogens are common oxidizing agents.
What are examples of oxidizing agents?
Oxidants are generally in the highest possible oxidation state because they tend to accept and reduce electrons. Some examples of oxidizing agents are halogens, potassium nitrate, fluorine, chlorine and nitric acid, etc.
Which is the strongest oxidizing/reducing agent?
Therefore, lithium metal is the strongest reducing agent (most easily oxidized) among alkali metals in aqueous solution. The standard reduction potentials can be interpreted as the classification of substances on the basis of their oxidizing and reducing capacity.
What is meant by a strong oxidizing agent?
Strong oxidizing agent Pyridinium Chlorochromate (PCC) is a milder version of chromic acid. PCC oxidizes alcohols at one level of the oxidation ladder, from primary alcohols to aldehydes and secondary alcohols to ketones. Unlike chromic acid, PCC does not oxidize aldehydes to carboxylic acids.
What is a " good oxidizing agent" mean formula
Yes, you are right. The oxidizing agent absorbs electrons and forms more negatively charged particles. The oxidizing agent is reduced and the reducing agent is oxidized. Think of the expression LEOGER. they lose oxidized electrons, they gain reduced electrons.
What makes a class oxidizing agent dangerous?
Oxidation class Due to their high oxygen content, they are often reactive substances. They can react with other flammable or combustible materials and be burned by the heat generated.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Which is the strongest oxidizer in the world?
Which is the strongest oxidizer in the world?
Posted Oct 24, 2005 With electrolysis you can make oxidizing and reducing agents as powerful as you want. Fluorine is the strongest known chemical oxidizing agent and cesium is (probably) the strongest known chemical reducing agent, but electrolysis can create even more oxidative and reducing conditions.
Which is the strongest reducing agent in the world?
The strongest reducing agents are found in the corner of the table where metallic sodium and potassium are listed. Arrange the following oxidants and reducing agents in ascending order of concentration: Oxidizing agents: Cr 3+, Cr 2 O 72, Cu 2+, H+, O 2, O 3 and Na+.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is a " good oxidizing agent" mean for company
What is a " good oxidizing agent" mean for company
A commercial offer (or offer) is a document in which you describe the requested product or service and inform your buyer of the corresponding prices. In most cases, this is in response to an invitation to bid when a third party asks for your service or product.
What does it mean when a customer asks for a quote?
The budget is an official document. Explain your price for the job or order and give your customer a clear cost of the job. When a client asks you for a quote, it means they are seriously considering working with you.
![:brown_circle: :brown_circle:]() How to get a price quote accepted by a customer?
How to get a price quote accepted by a customer?
Here are five tips to help you convince customers to accept your offer: Make a good impression. Be clear, professional and attentive. You can surprise your customer by adding a little more value than you expected. Add a call to action at the end with a simple underwriting process.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() How to create a quote for a client?
How to create a quote for a client?
Offer Text - Describe the goods or services offered and provide pricing information. Offer Footer - Contains the total of all items, the tax amount, and the offer expiration date. Offer a call to action, such as B. your signature. For examples of the structure, see the models by following the link above.
What is a " good oxidizing agent" mean price
A proposal is a call to action. The proposal is an official document. Explain your price for the job or order and give your customer a clear cost of the job. When a client asks you for a quote, it means they are seriously considering working with you.
How to prepare a price quote for a new business?
You can use good accounting software to make professional estimates with all the information you need. You can start a new business with effective offers. Here are five tips to help you convince customers to accept your offers: Make a good impression. Be clear, professional and attentive.
![:brown_circle: :brown_circle:]() What does the quoted price mean on the NYSE?
What does the quoted price mean on the NYSE?
Some of the largest exchanges in the world include the New York Stock Exchange (NYSE), Nasdaq, the London Stock Exchange (LSE), and the Tokyo Stock Exchange (TSE). The bid price is the final agreement between the buyer and seller or the bid and ask price.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What's the difference between oxidizing agent and species oxidized?
What's the difference between oxidizing agent and species oxidized?
Click to enlarge Oxidized particles are atoms that oxidize (oxide loses electrons), and an oxidizer is one that oxidizes, so the oxidant is reduced.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Which is a better oxidant H2O2 or water?
Which is a better oxidant H2O2 or water?
Yes, H2O2 is a better oxidizing agent than water. No, this is not the standard relegation potential. Reason: In fact, the stability of H2O2 is very low due to the presence of an OO bond or a peroxide bond as shown in the structure. Therefore, H2O2 reacts with O2 to stabilize and make the next reaction thermodynamically stable.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is a " good oxidizing agent" mean based
What is a " good oxidizing agent" mean based
What is the main definition of quote? Quoting means repeating exactly the words of the speaker or author. A quote is also such a recurring passage or statement. To quote means to quote something as proof.
Why are substances with positive ORP readings called reducing agents?
Since substances with a positive redox potential want to take up electrons, they are considered oxidizing agents. On the other hand, electrons with an excess of ions can afford to lose ions due to the oxidizing agent without destabilizing. That is why they are called antioxidants or reducers.
Why does oxidising power increase from left to right?
Oxidizing power is the tendency to hold an electron in such a way that it moves from left to right over time.
Why are quats not a good disinfectant?
Because while quats kill germs on surfaces, home studies of quats have never shown that you or your family are healthier than using soap and water. In fact, no studies have shown that the incidence of illness at home is reduced when antibacterial cleaning products are used.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is a " good oxidizing agent" mean example
What is a " good oxidizing agent" mean example
In the presence of a strong electron donor (strong reducing agent), water acts as an oxidizing agent. In the presence of a strong electron acceptor (strong oxidizing agent), water serves as a reducing agent.
Why is water considered a poor reducing agent?
Since the oxidation potential is negative, water does not want to oxidize spontaneously and therefore water is also a bad reducing agent. Always remember that oxidants are reduced and reducing agents are oxidized.
Which is an example of a mixed quotation?
A mixed quote is an indirect quote that contains a directly quoted sentence (in many cases a single word or short sentence):.
What is a " good oxidizing agent" mean to kill
The oxidizer turns organic waste into a harmless gas that is released into the atmosphere. Basically, disinfectants kill germs, bacteria and disease. Oxidizing agents are used to remove non-bacterial contaminants and organic matter.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What's the difference between an oxidizer and a shock?
What's the difference between an oxidizer and a shock?
A disinfectant is used to kill pathogenic microbes. Sanitizing the water removes bacteria from the hot tub. On the other hand, the oxidizer/shock does not kill bacteria and is not a disinfectant. The oxidizer uses oxygen to absorb the organic waste that swimmers bring to the spa water.
What is the definition of a biocide agent?
DEFINITIONS "Biocide" is a general term describing a chemical agent, generally a broad spectrum, that inactivates microorganisms.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Can You give Me list of oxidizing agents?
Can You give Me list of oxidizing agents?
- fluorine. Fluorine is a Group VIIA compound commonly referred to as the halogen group.
- chlorine. Common chemical oxidizing agents Chlorine is a green member of group VIIA, not found freely in nature, but for example in combined form
- Bromine.
- Iodine.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What are some common oxidizing agents?
What are some common oxidizing agents?
The most common oxidizing agents are oxygen, hydrogen, ozone, potassium nitrate, sodium perborate, peroxides, halogens and permanganate compounds. Oxygen is considered the most common oxidizing agent.
What is an example of a reducing agent?
The reducing agent is normally in one of the lowest possible oxidation states and is known as an electron donor. Examples of reducing agents include earth metals, formic acid, oxalic acid and sulfite compounds.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Is Cl2 an oxidizing agent?
Is Cl2 an oxidizing agent?
Cl2 has gained an electron, which means it is a reduced substance. Br has lost an electron, meaning it is an oxidized substance. Therefore, Cl2 is an oxidizing agent and Br is a reducing agent.
Which one is a stronger oxidizing agent than zn
The standard reduction potentials can be interpreted as the classification of substances on the basis of their oxidizing and reducing capacity. Substances above H2 in Table 1 are stronger reducing agents (oxidize more easily) than H2. Substances below H2 are stronger oxidizing agents.
Which is the strongest oxidant in the table?
The strongest oxidizing agent in the table is F 2 with a standard electrode potential V. This high value corresponds to the high electronegativity of fluorine and tells them that fluorine has a stronger tendency to accept electrons (it is a stronger oxidizing agent) than any other.. another article.
![:brown_circle: :brown_circle:]() How is the charge of an oxidizing agent reduced?
How is the charge of an oxidizing agent reduced?
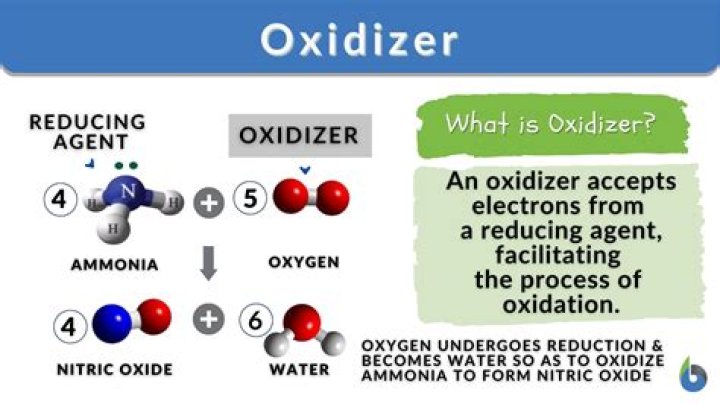
In an oxidation half reaction, the oxidizing agent is reduced because the absorption of electrons by the reducing agent causes a decrease in the charge or oxidation state of one of the oxidation atoms. This can be explained by the following equation: 2Mg (s) + O 2 (g) → 2MgO (s).
Which is the most common oxidizing agent in a reaction?
Two substances work together in these reactions: a reducing agent and an oxidizing agent. The most common oxidizing agents are oxygen, hydrogen, ozone, potassium nitrate, sodium perborate, peroxides, halogens and permanganate compounds. Oxygen is considered the most common oxidizing agent.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Which one is a stronger oxidizing agent for alcohol
Which one is a stronger oxidizing agent for alcohol
When oxidizing alcohol, two types of oxidants are used: strong and mild. Sometimes the same alcohol gives different strong and weak oxidation products. The following reagents are used as strong oxidants in the oxidation of alcohol. How do you know if alcohol is oxidized?
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Can a secondary alcohol be oxidized by a mild oxidizer?
Can a secondary alcohol be oxidized by a mild oxidizer?
The oxidation of the secondary alcohol is mainly carried out by a strong oxidizing agent. Mild oxidants are rarely used. Due to the absence of hydrogen atoms on the carbinol carbon in tertiary alcohol, they do not oxidize. Are you now going to learn how to identify primary, secondary, and tertiary alcohols using a strong oxidizing agent?
How are alcohols oxidized to aldehyde, ketone, or carboxylic acid?
Secondary alcohols are oxidized to ketones by strong oxidizing agents and mild oxidizing agents. Depending on the oxidizing agent used, the oxidation product of alcohol is an aldehyde or carboxylic acid. When alcohols are oxidized to carboxylic acids by strong oxidizing agents, their color changes as follows.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Which is the stronger oxidizing agent in a sugar reaction?
Which is the stronger oxidizing agent in a sugar reaction?
All the carbon in the sugar is oxidized to carbon dioxide and a lot of energy is released. Oxidation levels in reactions with sugars Nitric acid is a stronger oxidizing agent. It oxidizes both the aldehyde group and one of the alcohol groups of glucose. Nitric Acid, ENT.
Which one is a stronger oxidizing agent than i2
The greater the attraction of electrons, the stronger the oxidizing agent. Therefore, the element with the highest electronegativity is the strongest oxidizing agent. They compare your standard reduction potential. For example, potassium permanganate and hydrogen peroxide are oxidizing agents.
![:brown_circle: :brown_circle:]() Why is iodine not a strong oxidizing agent?
Why is iodine not a strong oxidizing agent?
On the other hand, iodine is not the strongest oxidizing agent and does not cause other particles to lose electrons easily compared to the oxidation potential of chlorine, bromine or fluorine. Other halogens can also oxidize iodine anions, but not vice versa.
Which is a reducing agent and which is an oxidizing agent?
It is only in one electron, so it accepts one electron and is therefore an oxidizing agent. Reducing Agent: Substance that is reduced by oxidation and release of electrons. Here I have an extra electron. So if there is a substance that desperately needs electrons, it will leave them.
Which is the stronger oxidizing agent hydrogen peroxide or peroxide?
The one with the most positive reduction potential is the strongest oxidizing agent. Therefore, hydrogen peroxide is the most powerful oxidizing agent.
Which one is a stronger oxidizing agent for hand sanitizer
F2 is such a good oxidizing agent that metals, quartz, asbestos and even water ignite in its presence. Other good oxidizing agents are O2, O3 and Cl2, the elementary forms of the second and third most electronegative elements, respectively.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() When to use rubbing alcohol in hand sanitizer?
When to use rubbing alcohol in hand sanitizer?
Isopropyl alcohol is found in most hand sanitizers. Due to its strong bactericidal properties, isopropyl alcohol is used in hand and surface cleaners. It can also be used for household purposes. The surface to be disinfected should be cleaned with soap and water before applying isopropyl alcohol.
Which is more effective hypochlorous acid or disinfectant?
Hypochlorous acid is a weak acid with pK. at a temperature of about 25°C. Therefore, hypochlorous acid, the main disinfectant, predominates at lower pH levels and is a more effective disinfectant than hypochlorite ion, which has a higher pH.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Which one is a stronger oxidizing agent for essential oils
Which one is a stronger oxidizing agent for essential oils
Explanation: A typical table of standard reduction potentials is shown here. (Background) The species in the upper left corner have the highest reduction potential, making them the strongest oxidizing agents. The strongest oxidant on the list is F2, followed by H2O2, and so on up to the weakest oxidant, Li+.
Which is the strongest oxidizing agent in the world?
Ions, atoms and molecules with strong electron affinity are considered good oxidizing agents. The stronger the electron affinity, the greater the oxidative capacity. Elemental fluorine is considered the strongest elemental oxidizing agent.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What causes essential oils to be oxidized or no longer good?
What causes essential oils to be oxidized or no longer good?
Answer: Eventually all essential oils break down and the direct cause of this is a process called oxidation. The components of essential oils combine with oxygen from the air and cause negative chemical changes.
How do you rank oxidizing agents according to their reduction potential?
They classify oxidants according to their standard reduction potential. Below is a typical table of standard reduction potentials. The species in the upper left corner have the highest reduction potential, making them the strongest oxidizing agents.
Is HNO3 an oxidizing agent?
Yes, nitric acid (HNO3) is a very powerful oxidizing agent. The reason for this is that each nitric acid molecule contains 3 oxygen atoms. When an acid reacts with a metal, it forms a metal salt and hydrogen (H2) is released.
![:brown_circle: :brown_circle:]() Which is the best oxidizing agent in the world?
Which is the best oxidizing agent in the world?
Common oxidizing agents are oxygen, hydrogen peroxide and halogenated oxidizing agents. ABNO · acetone · acrylonitrile · cerium (IV) ammonium nitrate · ammonium peroxydisulfate · 2-azaadamantane Noxyl The original answer: Which oxidants are the best? Halogens are the best oxidizing agents.
Which is an oxidizing agent in a chemical reaction?
An oxidizing agent is a substance that transfers at least one electronegative atom to a chemical in a chemical reaction. The reducing agent (reducing agent) loses electrons and is oxidized in a chemical reaction. It is generally in one of the lowest possible oxidation states and is known as an electron donor.
Which is the best oxidizing agent for spinal muscular atrophy?
Halogens are the best oxidizing agents. They have a higher electronegativity value (with fluorine =). Although their oxidizing power reduces the group, the elements of group VIIA can be considered together as the best oxidizing agents. The first symptoms of spinal muscular atrophy may surprise you.
What are some good oxidizing agents for hand sanitizer
Examples of halogens that are good oxidizing agents are iodine, bromine, chlorine and fluorine. As mentioned earlier, fluorine is considered to be the strongest elemental oxidizing agent due to its higher electronegativity.
Which is the best disinfectant for sterilization?
Hydrogen peroxide (H2O2) is a widely used biocide for disinfection, sterilization and antiseptics. It is a clear, colorless liquid commercially available in various concentrations from 3% to 90%.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Are there restrictions on the use of hand sanitizer?
Are there restrictions on the use of hand sanitizer?
: Restrictions on Hand Sanitizer Use: This is a cosmetic or personal care product that is safe for consumers and other users under normal and reasonably predictable use. Cosmetics and consumer products specifically defined by global regulations are exempt from SDS consumer requirements.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What are the different types of chemical disinfectants?
What are the different types of chemical disinfectants?
Chemical disinfectants. 1 alcohol. Overview. 2 chlorine and chlorine compounds. 3 formaldehyde. 4 glutaraldehyde. 5 hydrogen peroxide.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Which is the most widely used antiseptic and disinfectant?
Which is the most widely used antiseptic and disinfectant?
Chlorhexidine is probably the most widely used biocide in antiseptic products, especially in mouth and hand rinses, and as a disinfectant and preservative. This is mainly due to the broad spectrum of action, the skin resistance and the low irritation.
What are some good oxidizing agents for ■■■■■■■■■
Good oxidants are also suitable for compounds with unusually high oxidation states such as permanganate (MnO 4), chromate (CrO 4 2) and dichromate (Cr 2O 7 2), as well as nitric acid (HNO 3), perchloric acid (HClO 4). ). and sulfuric acid (H 2 SO 4).
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Which is the best oxidizing agent for MnO 4?
Which is the best oxidizing agent for MnO 4?
By releasing electrons, it reduces the MnO 4 -ion to Mn 2+. Atoms, ions and molecules with an unusually high affinity for electrons are often good oxidizing agents. For example, elemental fluorine is the most powerful oxidizing agent.
![:brown_circle: :brown_circle:]() What are some good oxidizing agents for water
What are some good oxidizing agents for water
For example, elemental fluorine is the most powerful oxidizing agent. F2 is such a good oxidizing agent that metals, quartz, asbestos and even water ignite in its presence. Other good oxidants are O2, O3 and Cl2, the elemental forms of the second and third largest electronegative elements, respectively.
What kind of oxidizer can you use in a swimming pool?
1 nitrates. Nitrates give sausages their characteristic red color. 2 hydrogen peroxide. Hydrogen peroxide is stable enough to be used as a disinfectant. 3 Pool chemicals. Chlorine keeps swimming pools healthy, but it can also be dangerous. 4 nitric acid. Polishes for brass and other metals contain strong oxidants.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Which is an example of an oxidizing agent?
Which is an example of an oxidizing agent?
Examples of halogens that are good oxidizing agents are iodine, bromine, chlorine and fluorine. As mentioned earlier, fluorine is considered to be the strongest elemental oxidizing agent due to its higher electronegativity. Oxygen is the element corresponding to atomic number 8 and is denoted by the symbol "O".
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() How are oxidizers used in the making of bread?
How are oxidizers used in the making of bread?
Oxidizing agents or oxidizing agents are used to improve the mixing and separating properties of the bread dough. They improve the regeneration of gluten, which allows to control the hardness and elasticity of the dough. The oxidants used as dough conditioners are indispensable when baking bread at high speed.
How are nitrates used as an oxidizer in cooking?
1 Use of nitrates. Nitrates, especially sodium nitrates, are used to cure meats and make sausages. 2 hydrogen peroxide as an oxidizing agent. Hydrogen peroxide is a safe oxidizing agent, whose main function as an oxidizer is to increase the burning rate of combustible materials. 3 Pool chemicals. 4 uses of nitric acid.
What are the most common reducing agents?
The most common reducing agents are metals because they tend to lose electrons in their reactions with non-metals. The most common oxidants are halogens such as fluorine (F2), chlorine (Cl2) and bromine (Br2), as well as some oxyanions such as permanganate….
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Which is the oxidizing and reducing agent?
Which is the oxidizing and reducing agent?
In oxidative and reductive chemistry, an oxidizing agent is a substance that is reduced by a chemical reaction, causing another substance to oxidize. Common oxidizing agents are oxygen (O2), from which the term "oxidation" is derived, hydrogen peroxide (H2O2), ozone (O3) and halogen gases.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Which best describes an oxidizing agent for hand sanitizer
Which best describes an oxidizing agent for hand sanitizer
What is an oxidizing agent? An oxidizing agent (often called an oxidizing agent or oxidizing agent) is a chemical that tends to oxidize other substances, causing the substance to oxidize and lose electrons.
Which is the best chemical for sterilization and disinfection?
Although alcohol formaldehyde is a chemical sterilant and formaldehyde is a highly effective disinfectant, the medical use of formaldehyde is limited by its irritating fumes and pungent odor, even in very small amounts (