O3 Polar Or Nonpolar - How To Discuss
O3 Polar Or Nonpolar
Is O3 polar or non-polar? Emergency!!?
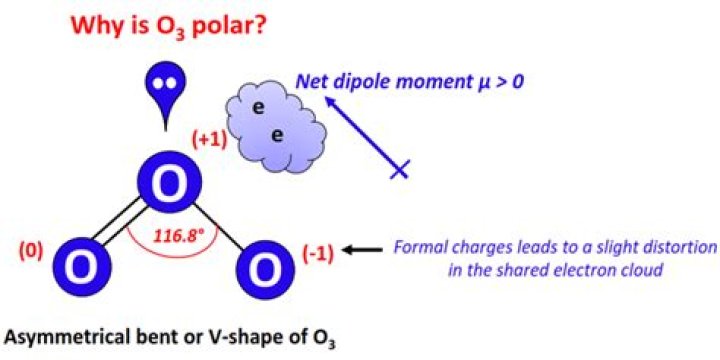
Well, ozone has two resonant forms, each with a double bond and each with a pair of non-closed electrons in the central atom, compressing the shape of the molecule into curved molecular geometry. As you know, curved geometry results in a liquid dopole moment and that is why ozone has a dopole moment and hence it is polar.
Dupole moments are a measure of vector, they have height and direction. Electro-negativity (size) is part of the problem with dupole moments, but so is the atomic position (direction). You are right that the electronegativity is the same for all three oxygen atoms, but the position of the oxygen atom also plays a key role in converting ozone into a polar molecule. So draw a Lewis structure, look at the differences in the electro-negativity of atoms, look at their positions and finally see if they cancel each other out.
Don't worry about doppel moments as most AP chemistry tests focus on reaction and balance. At least that was for me when I bought it 4 years ago. Anyway, okay.
O3 Polar Or Nonpolar
O3 Polar Or Nonpolar
Is O3 polar or non-polar? Emergency!!? 3
So my AP review do chemistry tomorrow, and it really bothers me that my review book only claims that O3 is polar. It is possible ? There can be no difference in electrical negativity between the same atoms in a molecule, right? I only thought that Doppol would be stronger moment by moment because of the number of electrons due to London's strong scattering force, but what is it? Sorry, I'm really under pressure and it bothers me a lot.
Ozone has two resonant shapes, each with a double bond and each with a pair of unconnected electrons in the central atom, compressing the molecular shape into curved molecular geometry. As you know, curved geometry results in a liquid dopole moment and therefore ozone has a dopole moment and hence it is polar.
Dupole Moment is a collagen vector, both of which have MAGNITUDE and DIRECTION. Electronegativity (size) is part of the dupole moment problem, but so is the atomic position (direction). You are right that the electronegativity is the same for all three atoms of oxygen, but the position of the oxygen atom also plays a key role in converting ozone into polar molecules. So draw a Lewis structure, look at the difference in the electrical negativity of atoms, look at their positions, and finally see if they cancel each other out.
Don't worry about doppel moments as most AP chemistry tests focus on reaction and balance. At least that's what happened to me when I bought it 4 years ago. Anyway, okay there.
This is O3 polar.
O3 Polar Or Nonpolar
O3 Polar Or Nonpolar
This page can help you.
D:
Is O3 polar or non-polar? Emergency!!?
So my review of APCam yesterday, and it really amazes me that my review book only claims that O3 is polar. It is possible ? There can be no difference in electrical negativity between the same atoms in a molecule, right? I thought it had a strong doppel ...


