Nh3 Molecular Geometry - How To Discuss
Nh3 Molecular Geometry
What is Electronic Geometry or Molecular Geometry of Ammonia (NH3)?
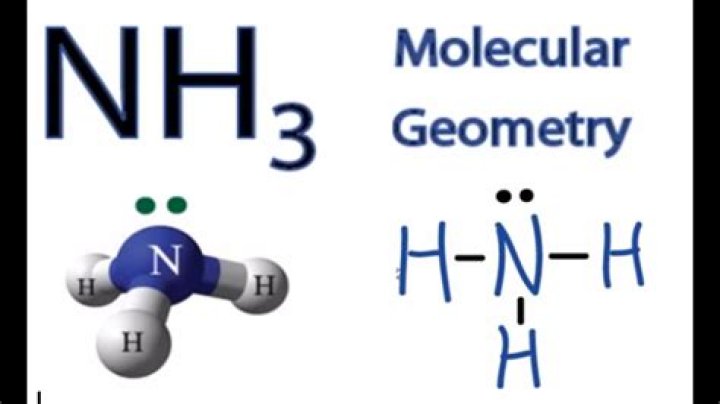
In ammonia, N has 3 bonds and one pair of electrons, forming a total of four bonds. Whereas, the geometry of the electron will be tetrahedral. Molecular geometry, on the other hand, has the shape of a triangular pyramid, as predicted by VSPR theory, because it has three pairs of electrons instead of four bonded atoms.
p which helps. : D.
Ammonia molecular geometry
NH3. Molecular geometry
This page can help you.
D:
What is Electronic Geometry or Molecular Geometry of Ammonia (NH3)?
Ammonia (NH3) has electronic geometry and molecular geometry
OK, here's the answer
The geometry of the electronic domain is linear
Molecular geometry is linear
Tetrahedron, Tribal Pyramid
Tetrahedron because it has four bound domains. (3 links + 1 isolated pair)
Pyramid triangle because it has non-binding domains
Nh3 Molecular Geometry
Nh3 Molecular Geometry
What is Electronic Geometry or Molecular Geometry of Ammonia (NH3)? 3
Ammonia has electronic geometry and molecular geometry (NH3).
In ammonia, N has 3 bonds and one pair of electrons, forming a total of four bonds. There, the geometry of the electron will be tetrahedral. Molecular geometry, on the other hand, has the shape of a triangular pyramid, as predicted by the VSPER theory, because it does not consist of four connected atoms, but three and one pair of electrons.
p which helps. : D
This page can help you.
D:
What is Electronic Geometry or Molecular Geometry of Ammonia (NH3)?
Ammonia has electronic geometry and molecular geometry (NH3).
Well here is the answer
The geometry of the electronic domain is linear.
Molecular geometry is linear.
Tetrahedron, triangular pyramid
Tetrahedron because it has four binding domains. (3 links + 1 isolated pair)
Pyramid Triangle because it has non-binding domains (isolated pairs)