Krf4 Lewis Structure - How To Discuss
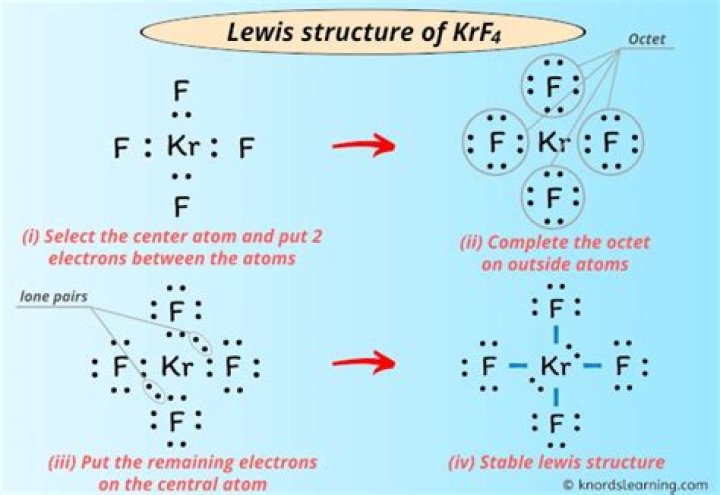
Krf4 Lewis Structure
What is the molecular structure and binding angle of KrF4? 3
I think the original VSPER array, which describes individual pairs and bonds, is octahedral, but the molecular structure cannot be square because there is only one pair. So I guess it could be a tetrahedral or bipramidal triangle? I have to figure out the angles and I'm not sure if I should consider Tark myself ... please help! Specifications are welcome! Thanks in advance =)
KrF4 has square plane molecular geometry. The electron group geometry is octahedral. There are 4 F bonds that subtract 4 electrons from 8 electrons of Kr. So there are 4 lone electrons and that means there are 2 pairs of electrons. So the angles are 180 and 90.
For the best answer, search this site for the correct answer! If you draw the Lewis structure, you will find that it is a non-polar tetrahedral molecule because the dopoles cancel each other out. give). The formal oxidation state of an atom in a molecule (or molecular ion) can be calculated as follows: (number of valence electrons in an independent atom) minus (half the number of electrons attached to that atom) minus (number of non-valence electrons split molecule) Of electrons attached to atoms. KrF4 is a polar molecule.> Absolutely wrong! It is a non-polar molecule because K4 has F and 2 isolated pairs. It is a square AX4E2, not polar. 4). The connection angle for IF6 + is 90. > Absolutely right! It is AX6, octahedral shape with all angles like 90 ° 5). SeF4 is a polar molecule. > Wrong Wrong. If you count the total number of valence electrons (34 v), you know that you have an isolated pair. So this is an AX4E molecule (in the form of lead). Due to the isolated pairs, dopoles do not cancel each other out, so it is a polar molecule. 6). The NO3 has three Lewis resonant structures. > Absolutely right! If you pull it, you will know that oxygen has a double bond. But which one As there are three, the double bond can be placed on any of the three O atoms, forming a 3-resonance structure. In fact, the link is between single link and double link.
Lewis structure of Krf4
Krf4 Lewis Structure
Krf4 Lewis Structure
1. True 2. True 3. False 4. True (octahedral structure) 5. True, seen with lone pairs around Se atoms. Geometry = Triangular Pyramid 6. True, we don't just agree with n. 5


