Iodine Electron Configuration - How To Discuss
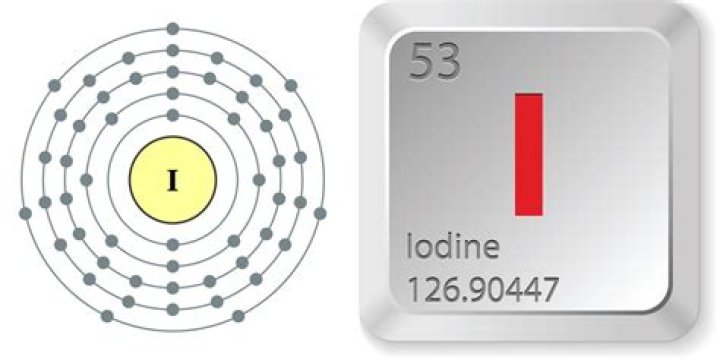
Iodine Electron Configuration
What is the electronic configuration of iodine?
As mentioned above for iodine, [Kr] 4d10 5s2 5p5
* Probability * Levels N = 5 are ■■■■ L = 0, 1, 2, 3, 4 OS, P, D and F. The largest atom ever discovered contains electrons in the formation of its earthly state at the 5f level.
At n = 6 level * possible * verbal exercises are l = 0, 1, 2, 3, 4 and 5 or s, p, d, f and (theoretically) g. None of the elements found so far have enough electrons for gortals in its ground state.
S is the highest principal quantum number in the formation of electrons. The energy of an electron is a function of its principal quantum number (n).
The order in which electrons are recharged (built) into atoms is not the order in which they are ionized (elimination of electrons). It is a painful point to ignore elementary chemistry courses.
Ogi involves assuming that you are on the dock, aboard a raft with many legs that they can climb freely. You'll climb to the top (hey, you're lazy) because it's the easiest to reach. But by doing so, you change the height (along with the raft and other occupants). The other side of the fence may be more now. So when it's time to disembark and head back to the pier, the second higher (closer) section goes down first, even if you come last.
This is not a big deal, but it does mean that the energy status of the atom is neither dense nor stationary and depends on the possession of the atom.
[CR] 4d10 5s2 5p5
All ■■■■ documents are blank and will remain blank most of the time.
The electron composition of I is: [Kr] 4d10 5s2 5p5
Iodine Electron Configuration
Iodine Electron Configuration
What is the electronic configuration of iodine? ۔
And what are the 5th and 6th levels of verbal energy?
Since iodine is at 5 in the table of elements, when its high energy level is only at 4, correct me if I am wrong.
As mentioned above for iodine, [Kr] 4d10 5s2 5p5.
At the level of possible verbal * n = 5 are l = 0, 1, 2, 3, 4 os, p, d and f. The largest atom ever discovered has an electron in the order of its ground state at the level of 5f.
At the level of n = 6, * possible * verbal exercises are l = 0, 1, 2, 3, 4 and 5 or s, p, d, f and (theoretically) g. None of the elements found so far have enough electrons for gortels in its ground state.
S is the largest principal quantum number in the electron configuration. The energy of an electron is a function of its principal quantum number (n).
The order in which electrons are recharged (built-up) in atoms is not the same as the order in which they are ionized (elimination of electrons). This is a painful point that introductory chemistry courses ignore.
Ogi involves assuming you are on the dock, on a multi-legged raft that they can climb, and freely. You'll climb to the top (hey, you're lazy) because it's the easiest to get. But by doing so, you (along with the raft and others) change the height. Maybe another part of the raft is higher now. So when it's time to get off the raft and go back to the dock, the upper (closest) part goes down first, even if it's the last time you come.
This is not a big deal, but it does mean that the energy state of the atom is neither dense nor stable and depends on the possession of the atom.
The electron configuration of I is: [Kr] 4d10 5s2 5p5.
Iodine Electron Configuration
Iodine Electron Configuration
What is the electron configuration of iodine? 3
And what are the 5th and 6th levels of verbal energy?
Since iodine is at 5 in the table of IC elements, while its highest energy level is only at 4 ... Oops, correct me if I'm wrong.
As mentioned above for iodine, [Kr] 4d10 5s2 5p5
Possible * orals * at level n = 5 are l = 0, 1, 2, 3, 4 os, p, d and f. The largest atom ever discovered has electrons in the ground state setting at 5f level.
At the level of n = 6 * possible * verbal exercises are l = 0, 1, 2, 3, 4 and 5 or s, p, d, f and (theoretically) g. None of the elements found so far have enough electrons for Gortelles in its ground state.
S is the most principal quantum number in the order of electrons. The energy of an electron is a function of its principal quantum number (n).
The order of filling (accumulation) of electrons in an atom is not the same as the order of ionization (removal of electrons). This is a painful point that introductory chemistry courses overlook.
An OGI involves assuming you are on the dock and navigating a multi-segment fleet that can go up and down freely. You will be climbing to the top (you are lazy) because it is the easiest to reach. But by doing so, you (along with the fleet and other occupants) change the height. Maybe another part of the fleet is higher now. So when it's time to get off the boat and go back to the dock, the top (closest) part goes down first, even if you are the last.
This is not a big issue but its purpose is to emphasize that the energy state of the atom is neither dense nor static and it does not depend on the possession of the atom.


