Electronegativity Trend - How To Discuss
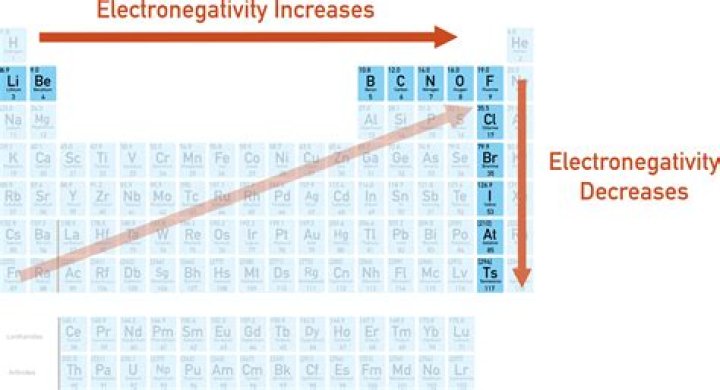
Electronegativity Trend
What is the trend of electronegativity on the periodic table?
In the periodic table, electronegativity usually increases when moving from left to right over time and decreases when descending from a group. Consequently, most of the electronegative elements are found in the upper right of the periodic table, while the less electronegative elements are found in the lower left.
Why does electronegativity in the periodic table increase over time?
In general, as the number of charges on the nucleus increases, electronegativity increases over time. It attracts the bonding electron pair more strongly. When you fall into a group, the electronegativity decreases as the bonding electron pair moves further and further away from the attraction of the nucleus.
So the question is, why is K greater than CA?
Potassium (K) has a larger atomic radius than calcium (Ca) because when we go down in a group the atomic radius increases and when we move a period from left to right the atomic radius decreases. The atomic radius of potassium (K) is greater than that of calcium (Ca).
A thousand thanks!Exactly what electronegativity trend do you see when you cross a certain period in the periodic table?
Moving from left to right over a period of time increases electronegativity, ionization energy and atomic radius decrease.
What are the causes of electronegativity?
Electronegativity increases as you move along the periodic table from left to right. This is due to a higher charge on the nucleus, which makes the electron bond pairs very attracted to the atoms that are more directly on the periodic table. Fluorine is the most electronegative element. Which element has the highest electronegativity?
Electronegativity increases in groups from bottom to top and from left to right over time. Therefore, fluorine is the most electronegative element, while francium is one of the least electronegative.
How do you explain electronegativity?
Electronegativity is a measure of an atom’s tendency to attract a pair of bonding electrons. The Pauling scale is the most used. Fluorine (the most electronegative element) is given a value of 4.0 and the values rise to Cesium and Francium, which are the least electronegative to 0.7.
Why is electronegativity a periodic trend?
The electronegativity of atoms increases over time in the periodic table from left to right. This is because if you walk left to right for a while, the nuclear charge increases faster than the electron shield, so the attraction of the atoms to the valence electrons increases.
What are the causes of low electronegativity?
Since the electron density of potassium is further away from the positive nucleus, protons have fewer properties. If an atom is larger and has more electronic shells, it has lower electronegativity, if the nucleus has fewer protons in the same time period, it has lower electronegativity.
What happens to the ionization energy over time?
If you move from left to right or up in a group over a period of time, the initial ionization energy usually increases. As the atomic radius decreases, it becomes more difficult to remove an electron closer to a positively charged nucleus. They experience a weaker attraction to the positive charge of the nucleus.
Is HCL polar or non-polar?
HCl is a polar molecule because chlorine has a higher electronegativity than hydrogen. It then attracts electrons to pass more time, which gives it a negative charge and hydrogen a positive charge.
How do you know if Br2 is polar or non-polar?
Which element has the highest ionization energy?
This trend shows that cesium has the lowest ionization energy and fluorine has the highest ionization energy (excluding helium and neon).
Is H2O polar or non-polar?
A water molecule, abbreviated to H2O, is an example of a polar covalent bond. Electrons are distributed differently and the oxygen atom spends more time with electrons than hydrogen atoms. As electrons spend more time with the oxygen atom, it carries a partially negative charge.
Which element has the second lowest electronegativity?
Francium has the lowest electronegativity value.
Why is oxygen so electronegative?
The oxygen atom is more electronegative (it attracts electrons better than hydrogen because it has more positively charged protons in the nucleus), and this makes it a little more negative so that the hydrogen atoms cannot hold electrons and get a little more positive.
What is the most electronegative C or S?
Phosphorus and sulfur belong to the same time and are conceptually more electronegative than phosphorus. Carbon should be the most electronegative of the three.


