Clf4 Molecular Geometry - How To Discuss
Clf4 Molecular Geometry
What is the molecular geometry of ClF4 +? 3
Help
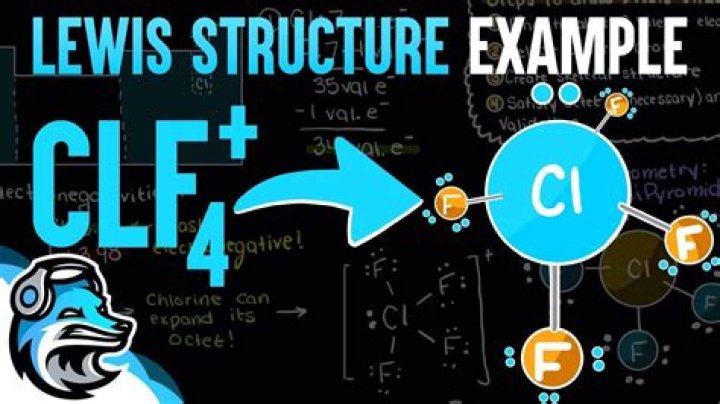
ClF4 + has 34 electrons. There are four pairs of Cl bonds for four F atoms. Each F with one electron byte which will represent 32 electrons. Finally, we need to add the remaining pairs of electrons to the central atom of Cl. This gives Cl five pairs of electrons, consisting of four bonding pairs and one isolated pair. Using VSEPR theory, it leads to a triangular pyramid electron pair geometry and a dual molecular geometry with isolated pairs in the equatorial position.
Lewis structure of Clf4
ClF41 + has 34 valence electrons. Cl has 4 binding pairs around and 1 disconnected pair. If all 5 electron groups are included in the bond, the shape will be a triangle-pyramid. With non-sticky mate, the shape becomes rocky.
This page can help you.
D:
What is the molecular geometry of ClF4 +?
Help
See my link
Clf4 Molecular Geometry
Clf4 Molecular Geometry
This is a square project.