Ch3 Lewis Structure - How To Discuss
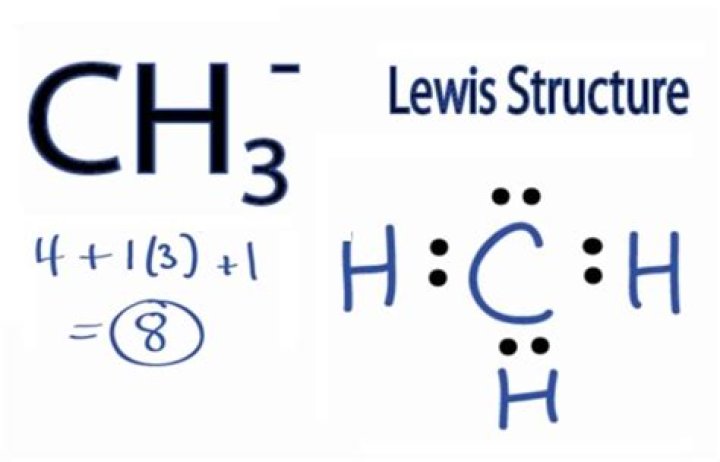
Ch3 Lewis Structure
Are you going to develop the Lewis structure of (CH3) 2S? Does it have 20 valence electrons?
A molecule does not have valence electrons, although it can have a pair of non-binding electrons, if you mean that.
Count the number of first electrons:
2 * 4 [2 for carbon] + 6 * 1 [6 for hydrogen] + 6 [for sulfur]
= Total 20 electrons. Still with me
You just have to be more discriminating with the help you render toward other people. Sulfur appears to be the central atom with 2 CH3 groups attached. Since HC bonds will be individual (because of H and because there should be room for S bonds) and this also means that SC bonds must be individual, all molecules are single bonds.
S with 2 pairs of isolated electrons.
And yes, it looks like everyone has a byte (and a pair of H, 2 electrons because it's special :)). If you count the electrons, it should be 20, which is also the case. (The bond counts as two electrons, so 8 * 2 + 4 = 20).
For example, I help.
0. Chemical compound = (CH3) 2S
1. Count the number of valence electrons in your chemical compound.
Walk. = 2 (4) [carbon] + 2 (3) [hydrogen] + 1 (6) [sulfur] = 20
2. Our chemical compound tells us that two CH3 molecules are connected to the siller element.
You put it in your Sulr and connect the carbon left and right. Then add 3 hydrogen atoms to each carbon.
You use 16 EVs or [8 Link X2 EVs]. So you currently have 4 or [(20 16) EV] left.
They know that hydrogen needs only 2 electrons to be happy and that we have the most electrical element. So he put the other 4 EVs on the cylinder.
H.H.
| .. |
HCSCH
| .. |
H.H.
Ch3 Lewis Structure
Ch3 Lewis Structure
Are you going to pull the Lewis structure of the (CH3) 2S? Does it have 20 valence electrons? ۔
A molecule does not have valence electrons, although it may have a pair of electrons, if that is what you mean.
Count the number of first electrons:
2 * 4 [2 for carbon] + 6 * 1 [6 for hydrogen] + 6 [for sulfur]
= Total 20 electrons. Still with me
You just have to be more discriminating with the help you render toward other people. Sulfur appears to be the central atom with 2 CH3 groups attached. Since HC bonds must be unique (because of H and because there must be room for S bonds) and it also means that SC bonds must be unique, molecules have all single bonds.
S with 2 pairs of isolated electrons.
And yes, it looks like everyone has a byte (and H has a pair, 2 electrons because it's special :)). If you count the electrons, it must be 20, which is also the case. (The bond counts as two electrons, so 8 * 2 + 4 = 20).
For example, I help.
0. Chemical compound = (CH3) 2S.
1. Count the number of valence electrons in your chemical compound.
Floor = 2 (4) [carbon] + 2 (3) [hydrogen] + 1 (6) [sulfur] = 20
2. Our chemical compound tells us that two CH3 molecules are connected to the Sulr element.
You put it in your cellar and joined the carbon left and right. Then attach each hydrogen atom to each carbon.
You use 16 EVs or [8 Link X2 EVs]. So you currently have 4 or [(20 16) EVs] left.
They know that hydrogen needs only 2 electrons to be happy and Sulr is our most electric element. So you put the other 4 EVs in Sulr.
H.H.
| .. |
HCSCH
| .. |
H.H.
CH3 + formal charge removed? ۔
CF3 + w Would you like to draw a Lewis Point diagram for this? If not for formal position then C 4 + 7 + 7 + 7 = 25.
251 = 24
I learned that carbon always has 4 bonds. My teacher drew with 3 links?
C around 3 F and only one link in each CF. Don't have 4 responsibilities? So the double bond in one of the F? The official value of C 4 (40) = 0 but then it is not true. Ah very simple.
CH3 + is very unstable because there is no way to fill a carbon byte when there are only 6 valence electrons in the whole molecule. This means that every hydrogen must have a bond, but the reason is that there are no other electrons.
CF3 + also has tse 3 bonds with other fluorine atoms, as no fluorine atom can have double bonds. Fluorine has no backing, so it cannot have more than one bond with one atom. Therefore, carbon 8 contains only 6 of the valence electrons.
It is more common to find CH3 (Idea ion) or CF3 (Trichloride ion) because they have a pair of carbon and 2 extra electrons to provide a full byte. However, it is also very rare because CH4 and CF4 are very stable compounds.
Ch3 Lewis Structure
Ch3 Lewis Structure
Are you going to pull the Lewis structure of (CH3) 2S? Does it have 20 valence electrons? 3
A molecule does not have valence electrons, although it may have a non-binding pair of electrons, if that is what you mean.
Count the number of first electrons:
2 * 4 [2 for carbon] + 6 * 1 [6 for hydrogen] + 6 [for sulfur]
= Total 20 electrons. Still with me
You just have to be more discriminating with the help you render toward other people. Sulr appears to be the central atom to which 2 CH3 groups are attached. Since HC bonds must be unique (because of H and there must be room for S bonds) and this also means that SC bonds must be unique, the molecule contains all the single bonds.
S with 2 pairs of isolated electrons.
And yes, it looks like everyone has a byte (and H has a pair, 2 electrons because that's special :)). If you count the electrons, it should be 20, which is also the case. (The bond counts as two electrons, so 8 * 2 + 4 = 20).
For example, I help.
0. Chemical compound = (CH3) 2S
1. Count the number of valence electrons in your chemical compound.
Floor = 2 (4) [carbon] + 2 (3) [hydrogen] + 1 (6) [sulfur] = 20
2. Our chemical compound tells us that two CH3 molecules are connected to the Sulr element.
You put in your Sulr and connect the carbon left and right. Then add 3 hydrogen to each carbon.
You use 16 EV or [8 Link x 2 EV]. So you currently have 4 or [16 (20) EVs) left.
They know that hydrogen needs only 2 electrons to be happy and Sulr is our most electronegative element. Then you put the other 4 EVs in Sulr.
H.H.
| .. |
HCSCH
| .. |
H.H.
CH3 + formal charge removed? 3
CF3 + Would you like to outline Lewis Point for that? If the formal position is not for C 4 + 7 + 7 + 7 = 25.
251 = 24
I learned that carbon should always have 4 bonds. My teacher developed with 3 links?
3 Fs around C and only one link on each CF. Aren't there 4 obligations? So the double bond in one of the F's? So the formal charge for C4 (40) = 0, but that's not true ... ahhh that makes sense
Ch3 Lewis Structure
Ch3 Lewis Structure
CH3 + is very unstable because when there are only 6 valence electrons in the whole molecule, there is no way to fill the carbon byte. This means that there must be a bond with each hydrogen, but this is because there are not many valence electrons.
CF3 + has only tse 3 bonds with other fluorine atoms, as no fluorine atom can have double bonds. Fluorine has no backing, so it cannot have more than one bond with another atom. Therefore, carbon has only 6 out of 8 valence electrons.
It is more common to look for CH3 (Idea ion) or CF3 (Trichloride ion) because they contain a pair of carbon and 2 extra electrons to give a full byte. However, it is also quite rare because CH4 and CF4 are very stable compounds.