Calcium Valence Electrons - How To Discuss
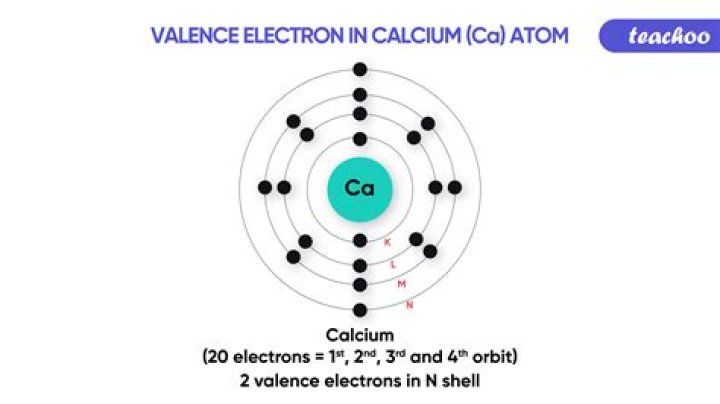
Calcium has 2 valence electrons. You can see that there are 2 electrons within the outer electricity degree and those are the valence electrons.17
Electrons and neutrons in a Calcium atom
The nucleus is located within the center of the atom. Protons and neutrons are located inside the nucleus. The atomic quantity of calcium is 20.
The atomic quantity is the wide variety of protons. That is, the range of protons inside the calcium is twenty.
Electrons same to protons are placed in a circular shell outside the nucleus. That is, the calcium atom has a complete of twenty electrons.
The number of neutrons in an element is acquired from the difference between the number of atomic hundreds and the wide variety of atoms. That is, neutron wide variety (n) = atomic mass number (A) – atomic number (Z)
We know that the atomic variety of calcium is 20 and the atomic mass number is ready forty(40.08u). Neutron (n) = 40 – 20 = 20. Therefore, the wide variety of neutrons in calcium(Ca) is 20.
Valence electrons of calcium(Ca)
The 3rd detail in organization-2 is calcium. All the elements of group-2 are referred to as alkaline earth metals. Therefore, calcium is an alkaline earth metal.
The valence electron is the entire range of electrons inside the final orbit. The total variety of electrons within the last shell after the electron configuration of calcium is known as the valence electrons of calcium(Ca).
The valence electrons determine the houses of the detail and take part within the formation of bonds.
Valence electrons of calcium
How do you calculate the quantity of valence electrons in a calcium(Ca) atom?
The valence electron needs to be decided by means of following a few steps.
The electron configuration is one among them. It isn’t always feasible to determine the valence electron with out electron configuration.
Knowing the electron configuration within the proper way, it is very smooth to determine the valence electrons of all of the elements.
However, valence electrons may be effortlessly recognized by arranging electrons consistent with the Bohr principle. Now we can learn how to determine the valence electron of calcium(Ca).
Step-1: Determining the entire quantity of electrons in calcium
1st we want to realize the overall variety of electrons in the calcium atom. To realize the range of electrons, you want to recognize the range of protons in calcium.
And to recognize the wide variety of protons, you need to understand the atomic variety of the calcium element.
To realize the atomic quantity we want to take the help of a periodic desk. It is essential to recognise the atomic quantity of calcium(Ca) elements from the periodic table.
The atomic number is the wide variety of protons. And electrons identical to protons are placed out of doors the nucleus.
Position of calcium(Ca) inside the periodic table
That is, we can in the end say that there are electrons same to the atomic range within the calcium atom.
From the periodic table, we see that the atomic wide variety of calcium(Ca) is 20. That is, the calcium atom has a complete of twenty electrons.
Step-2: Need to do electron configuration of calcium
Step 2 is very critical. In this step, the electrons of calcium(Ca) have to be organized. We know that calcium atoms have a total of twenty electrons.
The electron configuration of calcium suggests that the first shell of calcium has two electrons, the second one shell has eight electrons, the 3rd shell has 8 electrons and the 4th shell has two electrons.
The electron configuration of calcium thru the sub-orbit is 1s2 2s2 2p6 3s2 3p6 4s2.
Step-3: Determine the valence shell and calculate general electrons
The 1/3 step is to diagnose the valence shell(orbit). The remaining shell after the electron configuration is called the valence shell.
The general number of electrons in a valence shell is known as a valence electron. The electron configuration of calcium indicates that the remaining shell of calcium has two(4s2) electrons. Therefore, the valence electrons of calcium(Ca) are two.
Valence Proton for Calcium (Ca)
The potential of one atom of an element to join some other atom at some point of the formation of a molecule is called valency(valence).
There are a few policies for diagnosing valency. The number of electrons in an unpaired nation in the closing orbital after the electron configuration of an atom is known as the valency of that element.
Calcium donates two electrons of the remaining shell to shape a bond. Therefore, the valency of calcium is two.
Valency and valence electrons of calcium
The elements that have 1, 2, or 3 electrons inside the final shell donate the electrons within the ultimate shell at some stage in bond formation.
The factors that shape bonds via donating electrons are called a cation. Calcium donates electrons of the closing shell to form a bond and change into a calcium ion(Ca+2). That is, calcium is a cation detail.
Ca – 2e– → Ca+2
The electron configuration of calcium ion(Ca+2) is 1s2 2s2 2p6 3s2 3p6. The electron configuration of calcium ion(Ca+2) shows that calcium ion has three shells and the remaining shell has eight electrons(3s2 3p6).
This electron configuration suggests that the calcium atom has acquired the electron configuration of argon.
In this case, the valency of the calcium-ion is +2. Since the remaining shell of a calcium-ion has eight electrons, the valence electrons of calcium-ion(Ca+2) are 8.
Compound formation of calcium
The valence electrons take part in the formation of bonds with atoms of different factors. The electron configuration of oxygen shows that the valence electrons of oxygen are six.
The calcium atom donates its valence electrons to the oxygen atom and the oxygen atom gets those electrons.
As a result, oxygen acquires the electron configuration of neon, and calcium atoms collect the electron configuration of argon.
Calcium oxide(CaO) is shaped via the alternate of electrons among one atom of calcium and one atom of oxygen. Calcium oxide(CaO) is an ionic compound.
In the query it is requested how many valence electrons are present in a Calcium (Ca) atom. From the decrease instructions we’re managing specific elements and its position inside the periodic desk.
We recognise that the factors in the periodic desk are arranged in line with the growing order of the atomic range. So as the atomic variety increases the range of electrons inside the atom is also increasing.
The atomic quantity of an detail is equal to the quantity of electrons gift inside the atom.
So now permit’s talk the placement of Ca atom in the periodic desk, the atomic quantity and approximately the valency shown by means of Calcium.
The calcium is present in the left facet of the periodic table, with the alkaline earth steel agencies i.E. The group-2 elements and it’s far present inside the fourth period of the organization.
While reading about the periodic desk we’ve got studied about the shells and orbitals and we recognize that during a group as we move down a brand new shell is introduced.
The duration wide variety might be same to the entire number of shells gift in the atom.
As the Ca is present within the fourth duration, there are 4 shells gift in the Ca atom. And the atomic quantity of Ca is 20, subsequently there are 20 electrons in the atom.
Now let’s write the electronic configuration of Ca-
1s22s22p63s23p64s21s22s22p63s23p64s2
We realize that the valence electrons are the ones that are gift inside the outermost shell of the atom. From the digital configuration we know that the outermost shell of Ca is the fourth shell and the quantity of electrons in that shell is the valence electrons.
Here there are two valence electrons inside the outermost shell.
Note: The valence electrons debts for the valency of the atom i.E. The valency of the atom is the range of electrons that can be shared or eliminated in the chemical response to form the chemical bonds.
These electrons inside the outermost shells are the electrons which actively take part in the bond formation. The valency of Ca is +2, i.E. Two electrons may be donated via the atom for the chemical response.
Check out the blackboard. That box on the left has all of the statistics you need to recognise about one element. It tells you the mass of 1 atom, what number of pieces are interior, and in which it have to be positioned on the periodic desk.
Now we’re operating with the fourth duration/row in the desk of factors. You may additionally have an easy manner to know the variety of electrons in a impartial atom, however the placement of those electrons receives a little extra complicated.
Let’s take a look at the preparations of electrons inside the simple elements (left and right sides of the table) of length 4 and the extra complicated preparations of the transition elements (within the middle of the row). If you watched this is a bit over your head, cross returned and examine the elements 1-18 which have companies which can be a little extra simple.
Electrons In The Shells
Take a observe the image underneath. Each of those coloured balls is an electron. In an atom, the electrons spin across the middle, also referred to as the nucleus. The electrons want to be in separate shells/orbitals.
As you study more about atomic shape, you’ll research that the electrons don’t live in described areas across the nucleus.
They are observed in clouds which can have exceptional shapes that include spheres and dumbbell-like shapes. So don’t forget whilst you take a look at our breakdown that the electrons are not usually in a nice neat order as proven right here.
You will locate calcium within the 2d column of the duration desk with different elements including beryllium and magnesium.
Those factors make up the alkali earth metal circle of relatives. Yes, calcium is described as a metallic because of both its bodily and chemical traits.
They all have an outer shell with two electrons and are very reactive. Those factors in the second column have electrons ready to make compounds. It shouldn’t wonder you that calcium has a valence of 2.
You might not discover calcium sitting round as a natural detail. In nature, calcium is usually determined in compounds with different factors. One of the maximum famous compounds is limestone (CaCO3).
Summary
These electrons inside the outermost shells are the electrons which actively take part in the bond formation. The valency of Ca is +2, i.E. Two electrons may be donated via the atom for the chemical response. Check out the blackboard. That box on the left has all of the statistics you need to recognise about one element. It tells you the mass of 1 atom, what number of pieces are interior, and in which it have to be positioned on the periodic desk.
Frequently Asked Questions
Why does CA most effective have 2 valence electrons?
The number of electrons in an unpaired state within the closing orbital after the electron configuration of an atom is known as the valency of that element. Calcium donates electrons of the final shell to form a bond. Therefore, the valency of calcium is 2.
Does calcium have 2 valence electrons?
A: Calcium is a group 2 element with valence electrons. Therefore, it is very reactive and gives up electrons in chemical reactions.
How do you determine valence electrons?
For impartial atoms, the wide variety of valence electrons is identical to the atom’s primary group variety. The most important group variety for an element may be determined from its column at the periodic table. For example, carbon is in institution 4 and has four valence electrons. Oxygen is in group 6 and has 6 valence electrons.
How is the valency of calcium 2?
Explanation: calcium is in group 2 so its valency is valency is the number of outer shelll electrons it desires to have an solid outer shell . It needs 2 extra .
How many valence electrons does a Ca 2+ ion have?
A calcium 2+ ion has lost its two valence electrons, and now has 18 electrons. The electron configuration of a Ca2+ ion is 1s22s22p63s23p6 , that’s isoelectronic with the noble gasoline argon.
How many valence does calcium have?
2 valence electrons
Calcium has 2 valence electrons. You can see that there are 2 electrons in the outer energy degree and those are the valence electrons.
How many electrons does calcium have?
Twenty electrons
These are in four energy stages (1 although four) and it has the subsequent electron configuration. There are 2 electrons on the primary energy level, 8 electrons on the second level, eight electrons on the third strength degree, and 1 on the fourth energy level. That is, the calcium atom has a complete of twenty electrons.
How many valence electrons are in a calcium atom and a calcium ion?
Valence electrons
From the digital configuration we understand that the outermost shell of Ca is the fourth shell and the number of electrons in that shell is the valence electrons. Here there are valence electrons inside the outermost shell. So the wide variety of valence electrons in Ca is 2.
What is the electron distribution of calcium?
The variety of electrons for the Ca atom is 20. Therefore the Calcium electron configuration will be 1s22s22p63s23p64s2. The first electrons of calcium will cross in the 1s orbital. The subsequent 2 electrons for Calcium cross inside the 2s orbital.
How many valence electron does antimony have?
Antimony includes 5 electrons in its outer shell, accordingly it has 5 valence electrons. The digital configuration of Antimony is 2, 8, 18, 18, 5.
How many protective electrons does calcium have?
The Zeff of Ca is two.85 Explanation of values: Calcium has 10 electrons which can be taken into consideration internal core, n-2 or lower in electricity. These are the two electrons from n=1, and 8 electrons from n=2. There are eight electrons from n=3, but these are n-1, so count number for 0.Eighty five defensive.
Conclusion
At the end of this article hopefully you will like reading this article this is our great research on this topic. So if you will like reading this article please share this article to all your friends and others.
Related Article
Calcium Valence Electrons
Does calcium cation (Ca +) have a total number of electrons and many valence electrons? 3
I borrowed it with cash.
Ca2 + has a total of 18 electrons (I don't know if Ca + is, if so it would be 19, with 1 valence electron)
Ca2 + has z valence electrons.
It has 20 electrons and 2 valence electrons.
Ca2 + has 18 electrons and 0 valence electrons.
Ca + (the youngest node) will have 19 electrons.


