Bf4 Lewis Structure - How To Discuss
Bf4 Lewis Structure
The following molecule / ion (a) CS2 (b) BF4 ”(c) Prepare the Lewis structure of HNO2, bonds NO (d) OSCl2, S are central atoms?
Lewis structure
CS2 ...... S = C = S ......... with four points each January
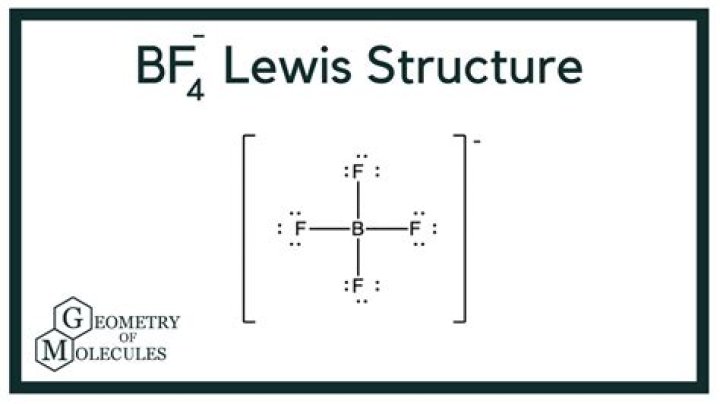
BF4 ...... A single bond of B on each of the four fluorine atoms, each fluorine has six additional points
Hà ˆÂ'Oà ˆÂ'N = O ........... Atoms N and O all have one byte of electrons
Oà Ã'Sà ˆÂ'Cl
...... |
Total ............... Each atom has one electron byte
Bf4 Lewis Structure
Bf4 Lewis Structure
Draw the Lewis structure of the following molecule / ion (a) CS2 (b) BF4 "(c) HNO2, of bond order NO (d) OSCl2, central atom S?
Lewis's structure
CS2 ...... S = C = S ......... with four dots in each cell
BF4 ...... A single bond of B in each of the four fluorine atoms, each fluorine has six extra points
HÃ Â'OÃ Â'N = O ........... Atoms N and O all have one byte of electrons
OÃ Â'SÃ Â'Cl
...... |
..... Cl ............... Each atom has one electron byte.