Sf2 Lewis Structure - How To Discuss
Sf2 Lewis Structure
What is the Lewis structure of SF2 1?
As far as I know, there are no stable SF2 ions. On the one hand, it will be free, meaning it has a strange number of electrons. The free S reacts strongly with a pair of closed electrons.
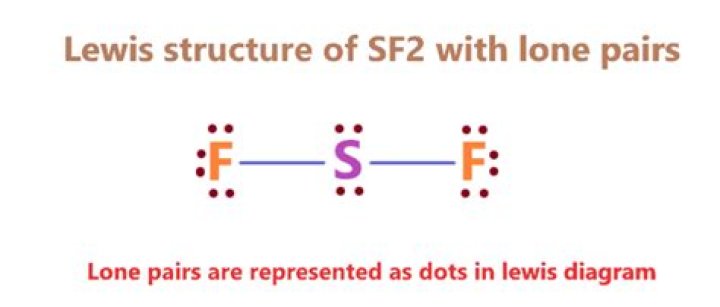
Fluorine has 7 valence electrons instead of six. There seems to be a problem with counting electrons. If Avon is present, it will have 21 electrons, six for sulfur and seven for fluorine, plus one due to charge. It will not have a double bond. Is a neutral sulfur fluoride molecule. It has a curved geometry with one bond on each fluorine and two isolated pairs on the sulfur.
Your potentially absent SF2 must have only one electron in the cell, which will give it an extra byte and make it an avon. This is not possible.
========= Tracking =========
His dream was a bit blurry to stretch Lewis's structure. You have to count the valence electrons to draw the Lewis structure correctly.
Hematites also have difficulty counting electrons. Question SF2 is 1, which I think is a charge of sulfur, two fluorines, and 1. Count the electrons: 6 + 2 (7) + 1 = 21.
Electro-negativity was first developed by Lens Palling. Like the Wolf Gang Paulie, Paulie was a proponent of quantum theory.
Lewis point structure for Sf2
Lewis structure of SF2
Sf2 Lewis Structure
Sf2 Lewis Structure
What is the Lewis structure of SF2 1? 3
So Sellar has 6 valence electrons and 2 fluorine has a total of 12 valence electrons = 18. And where every element has a lot of electrons (dots).
Basically what is the Lewis Framework?
As far as I know, there is no stable SF2 ion. On the one hand, it will be free, meaning it will have an odd number of electrons. The free S reacts considerably due to the single non-paired electrons.
Fluorine has 7 valence electrons instead of six. It seems that there is difficulty in counting electrons. If anion were present, it would have 21 electrons, six for sulfur and seven for fluorine, plus one charge. It will not have a double bond. Is a neutral sulfur di fluoride molecule. It has a curved geometry with a bond on each fluorine and two solitary pairs on sulfur.
Your potentially absent SF2 صرف only needs one electron, which will give you extended bytes and anionic bytes. This is not possible.
========= Tracking =========
His dream was a bit blurry to attract Lewis's structure. To draw the Lewis structure correctly, you must count the valence electrons.
Hemitetals also have difficulty counting electrons. Question SF2 is 1, which I think is a charge of sulfur, two fluorines, and 1. Count the electrons: 6 + 2 (7) + 1 = 21.
Electronegativity was first developed by Lens Paling. Like the Wolf Gang Paulie, Paulie was a proponent of quantum theory.
A cylinder has six valence electrons, while each fluorine actually has 7 valence electrons (this is a group of 7 atoms). That is, the number of electrons is 22 .... 2 (7) + 6 + 2 (for negative charge) = ??
Therefore, Sulr is the main atom in the structure with Noooo-Pi- (double) bonds. Think about it, fluorine is the most electronegative atom on the IC table. This means that it is holding its electrons very tightly. The presence of a double bond will give fluorine a regular charge of +1. This is not due to the fact that fluorine has a negative electrical potential of 4 on the poly scale.
So the central sulfur atom, a two sigma (simple) bond with fluorine, each has 3 sets of isolated pairs supporting our 16 electrons. The other 6 cylinders form three isolated pairs (which can exceed valence bytes because they are large atoms).
Sf2 Lewis Structure
Sf2 Lewis Structure
It is difficult to imagine the structure of the valence electron count. Instead, consider the properties of each atom. You have sulfur and 2 fluorides. Siller is in column 6 and all the atoms in column 6 form 2 bonds like oxygen. Fluorine forms bonds because it only needs one more electron to complete its ortho.
So you have one atom that makes 2 bonds and two atoms that make each bond.
The structure is F S F.
s has two pairs of electrons and each F has three pairs.
However, they claim that there is an additional charge for this molecule. As written, this is very unlikely, because no atom can carry extra electrons, but S2F 1 can be what you mean.
Suppose S2F, then the structure S S F is the first three pairs of electrons in a sulfur atom.
(Number of electrons in 24 ss of flood valence) (20 valence electrons) = Number of bound electrons 4/2 = 2 pairs of bonds. (Valence electron 20) (number of electrons in the outer atom ss 16 valence) = number of solitary electrons 4/2 = solitary pair of 2 electrons. The solution to this algorithm not only gives a Lewis point plot, but also answers 2 isolated pairs, i.e. cice 1 .. ..: FSF: is a reasonable representation haha ... ...
Discover the secret to creating realistic portraits with a pencil. This will help you reach the championship quickly.
All of these can be divided into 5 realistic resource design lessons.
+) w Realistic eye-catching
+) w A realistic sniffing
+) w Realistic mouth and tooth pulling
+) w Realistic listening
+) w Realistic hair pulling
Find realistic pencil portrait skills now!