Oxidation Number Of Calcium - How To Discuss
Oxidation Number Of Calcium
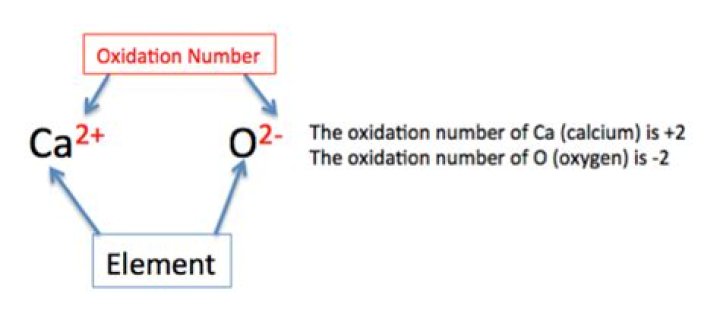
What are the oxidation numbers of calcium?
Since calcium has a value of 2+, its oxidation state is 2+.What is the oxidation number of oxygen?
The oxidation state of oxygen in compounds is 2, with the exception of peroxides such as H2O2 and Na2O2 where the oxidation state of O is 1. The oxidation state of hydrogen in compounds is +1, with the exception of metal hydrides such as NaH , LiH, etc., where the oxidation state of H is 1.Do you also know what the oxidation number of N is?
Nitrogen compounds, on the other hand, have nitrogen oxidation levels in the range from 3, as in the case of ammonia and amines, to +5, as in the case of nitric acid.What is the oxidation of C to CaCO3 in this way?
Carbon in CaCO3: The oxidation province of Ca is +2 and oxygen is -2.How is the oxidation state of an element determined?
Explanation:- The oxidation number of a free element is always 0.
- The oxidation number of a monatomic ion is equal to the charge of the ion.
- The oxidation number of H is +1, but when combined with smaller electronegative elements it is 1 inch.
- The oxidation number of O in compounds is generally 2, but 1 in peroxides.
What is the oxidation state of O to OH?
2What is the charge of CaCO3?
It is quite simple that the charge of the ion Ca is 2+ and the charge of the CO3 is 2, so the net sum of the charge of the compound CaCO3 + 2 + 2 = 0 (it is neutral). We can easily find the charge by adding the charges for each atom / molecule that is used to form the compound.What is the most common oxidation number for calcium?
2 Why does calcium have oxidation number +2? Rule 2 says that the oxidation number of a monatomic ion is equal to its charge. This rule applies to atoms that appear in substances such as monatomic ions. The charge is therefore about Ca +2. In fact, each calcium atom loses two electrons to produce Ca2 + ions and each O atom in O2 gains two electrons to produce O2 ions.What is the oxidation number of lithium?
Assign oxidation numbers to all elements of the following compounds: a) Lithium oxide Li2O O = 2. The molecule is neutral, so the lithium atoms should have a total oxidation number of +2. Since there are two, each must be +1.What is the oxidation state of chlorine in bleach powder?
In Ca (OCl) Cl bleach powder, the two Cl atoms have different oxidation states. In Fe3O4 the Fe atoms are found in two different oxidation states. Explanation: The formula for bleach is CaOCl2.What are oxidation numbers for?
Oxidation numbers are used by chemists to track electrons in a compound. We can use the guidelines to assign oxidation numbers to the atoms of a compound. Changes in the oxidation state during a reaction indicate an electron transfer.What is a good oxidant?
This means that they easily attract electrons. As you can see from the periodic table, the halogens that are good oxidants are fluorine, chlorine, bromine and iodine, with fluorine being the strongest oxidant of the four, followed by chlorine, bromine and iodine.How do oxidation numbers balance?
Redox Reaction Compensation: OxidationNumberChange Method Step 1: Assign oxidation numbers to each atom in the equation and write the numbers on the atom. Step 2: Identify which atoms have been oxidized and which have been reduced. Step 3: Use a line to connect atoms that are undergoing a change in oxidation number.What is the oxidation state of oxygen in peroxide?
1What are Fluorine Oxidation Numbers?
The oxidation number of fluorine (F) is always 1. The sum of the oxidation numbers of all atoms (or ions) in a neutral compound = 0.Can an oxidation number be a fraction?
In most cases, the atoms of the same element in a given compound have the same oxidation states, but each atom can have a different state. The individual oxidation numbers of an element in a compound can never be a fraction. However, the average oxidation number of an element in a compound can be a fraction.Oxidation Number Of Calcium
Oxidation Number Of Calcium
What is the oxidation state of calcium in calcium chloride? 3
And let me know if you know
The oxidation state of calcium is always 2+ because it loses two electrons from its outer s to reach the byte.
For chlorine it is always 1 .. All halogenes (Group VII) have one electron to get a byte because they have 7 electrons in their outer s.
Chloride Oxidation Index
Nothing to understand, just learn their names like history, biology, literature etc. Each compound has a unique name to distinguish it from others. Calcium chloride is not copper, just as a dog cannot tell a cat. It's very simple: you have to learn and know their names. Sulfuric acid (SO4) with two negative charges (2) Hydrochlorine (Cl) with one negative charge (1) Oxygen oxide (O) With two negative charges (2) Hydrogen (S) sulfide with two negative charges ( 2) Copper has two oxidation numbers (+1 and +2), copper (II) means you use +2 IRON has two oxidation numbers (+2 and +3), iron (II) means You use +2 calcium and magnesium has only one oxidation number (+2), you don't have to write it down. Sodium has only one oxidation number (+1), you do not need to write it if the total charge should be zero (initial positive charge + elemental negative charge = 0) copper (II) column Cu (+.) 2) + (SO4) (2), then Cu SO4 calcium chloride Ca (+2) + (Cl) (1) * 2, then Ca + (Cl) * 2 Ca Cl2 iron (II) oxide Fe (+2) + O ( 2), then Fe O copper oxide (I) Cu (+1) * 2 + O (2), then (Cu) * 2 + O Cu2O sodium sulfide Na (+1) * 2 + S (2), then ( Na) * 2 + S Na2 S Hydrate means that the molecule is lat (like the molecule of the surrounding water) mono = 1 //, di = 2 // tri = 3 // tetra = 4 // penta = 5 / / hex = 6 // hepta = 7, etc. Magnesium Sula Mg (+2) + SO4 (2), then Mg SO4 Magnesium Sula Heptahydrate Mg SO4 7H2O It took more time to write this exercise than learning anything else, suppose you know what to do from now on?
+2 because the oxidation state of calcium is +2. It belongs to group 2 in table ic and has 2s electrons.
Hello!!! Two Benefits Special principles apply to oxidation. For this I will mention at least two. This is the charge that contains the halide ion and, on the other hand, the charge of the electrically neutral substance. Remember its chemical formula is CaCl2. And halide ions are always negatively charged. For this reason, the halide ion is chlorine and note that the formula has its sub-2, which means that there are two chloride ions in one unit of the calcium chloride formula. Since there are two chloride ions, they obviously have two negative charges. However, the condition is that electrically neutral substances, such as pure elements and compounds (except the ions present themselves) have a net charge, ie we already have less than two, so we only need a useful charge of more than two. Need They net charge. Now, two good points, this is about calcium ions. So plus two plus minus two, which leads to net charge z and therefore fulfills this condition. I can help !!! Bless !!!


