Nh3 Ionic Or Covalent - How To Discuss
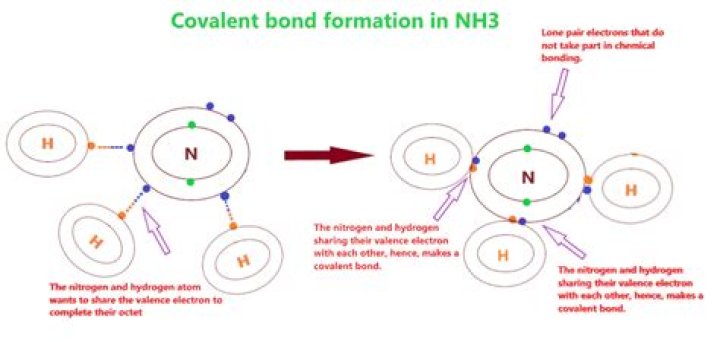
Nh3 Ionic Or Covalent
Is NH3 a covalent or ionic compound? Also read the details.
My friend said that the two elements on the right side of the table are aligned, and one on the left and one on the right. But ... since hydrogen is a gas ... but on the left and nitrogen on the right, is it ionic or coherent? I'm so stupid
Typically, metals come with non-metals through ionic bonds. Non-metals join other non-metals through covalent bonds.
An easy way to determine if a substance is compatible or ionic is to compare the electronegativity of the elements involved in the binding. Electronegativity is a measure of the attraction of electrons shared between atoms.
In NH3, the electronegativity of N is 3.04 and that of H is 2.20. If the difference in electro-negativity is less than 1.7, then the bond is compatible. Therefore, NH3 is free of cohesive bonds. But N is more attractive to share e than H, so nitrogen is partially negative and hydrogen is partially positive. This is called dopole and the bond is called polar covalent.
If two atoms have the same electro-negativity, then this bond is perfectly compatible. Some examples are H2, N2, O2, etc.
Is NH3 an ionic compound?
This is a general statement that applies to a connection that consists of two elements. Tses that make up ionic compounds usually consist of one element on the right (which makes cations = group 13) and one on the left (which makes ions, for example group 7) and the elements that make up ions They form a harmonious bond. This statement from the adjacent area is related to the proximity of atoms in the IC matrix and may contain anomalies.
In all cases, NH3 is compatible because the elements share electrons.
NH3 is definitely compatible.
Hydrogen is an element that is sometimes placed on the left and right side of the IC table. One of the disadvantages of the repeating table is that hydrogen does not really belong to any particular S. Many other shapes and designs try to correct this error. Think of hydrogen as the top of the pyramid, not the left.
NH3 compatible.
Nh3 Ionic Or Covalent
Nh3 Ionic Or Covalent
Is NH3 a covalent or ionic compound? Also read the details.
My friend said that the two elements on the right side of the table are covalent and one on the left and one on the right. But ... since hydrogen is a gas ... but nitrogen on the left and nitrogen on the right, is it ionic or covalent? I'm so stupid
Typically, metals come with non-metals through ionic bonds. Non-metals come in bonds with other non-metals.
An easy way to determine if a substance is compatible or ionic is to compare the electrical negativity of the elements in the bond. Electronegativity is a measure of the attraction of electrons shared between atoms.
In NH3, the electrical negative of N is 3.04 and H is 2.20. If the difference in electrical negativity is less than 1.7, then the bond is harmonized. Therefore, NH3 is free of covalent bonds. But N is more attractive to share e than H, so nitrogen is partially negative and hydrogen is partially positive. This is called dupole and the bond is called polar harmony.
If the electrical negativity of two atoms is the same, then the bond is purely compatible. Some examples are H2, N2, O2, etc.
This is a general statement that applies to a two-element connection. The ion compounds that make up Tse usually consist of the right side (cations = 13 groups) and the left hand (the formation of ions, such as group 7) elements and tse elements, which are aligned. Connection shapes are formed. This statement from the adjacent area is related to the proximity of atoms in the Matrix IC and may contain anomalies.
In all cases, NH3 is compatible because the elements share electrons.
NH3 is definitely compatible.
Hydrogen is an element that is sometimes placed to the left and right of the IC table. One of the disadvantages of repeated tables is that hydrogen does not really belong to any particular condition. Many other shapes and designs try to correct this error. Think of hydrogen as the upper part of the pyramid, not the left.
NH3 compatibility.


