Nh2cl Molecular Geometry - How To Discuss
Nh2cl Molecular Geometry
What is the molecular geometry of NH2Cl? 3
Tetrahedron as in ammonia.
Nh2cl Molecular Geometry
Nh2cl Molecular Geometry
This page can help you.
D:
What is the molecular geometry of NH2Cl?
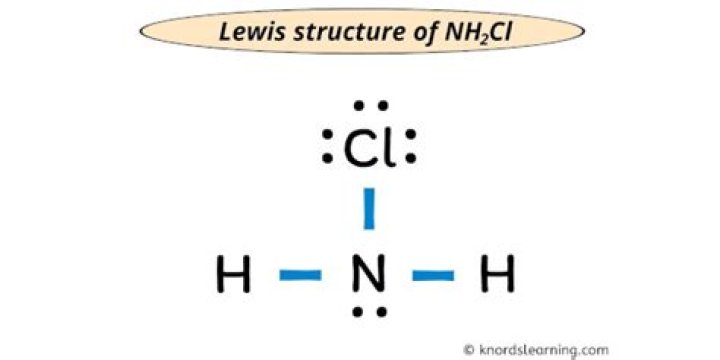
In fact, NH2Cl consists of an isolated pair and three atoms around the nitrogen atom. The resulting structure will be a triangular pyramid, not a tetrahedron.
Ammonia is surrounded by four H ions, while NH2Cl has only three surrounding ions and one pair. For this reason, NH2Cl is a triangular pyramid.
Bond angle NH3 (an H is replaced by Cl) would be similar to 107 degrees, if you do not understand these facts then know the bond angle of CH4 molecule which has no isolated pairs and 4 covalent bonds 109.5 degrees Is the angle of NH3 type molecules with 1 free electron pair and 3 covalent bonds have 107 degree bonds. H2O type molecule with 2 lone pairs and 2 covalent bonds has a bond angle of 104.5 degrees.