How Many Neutrons Does Potassium Have
How Many Neutrons Does Potassium Have
How many neutrons, protons and electrons are in a T-Til-atium?
Other than getting more points, it doesn't help much to say that now. But I will not do that.
IUMK
Serial No. 19
Atom meter 39.10
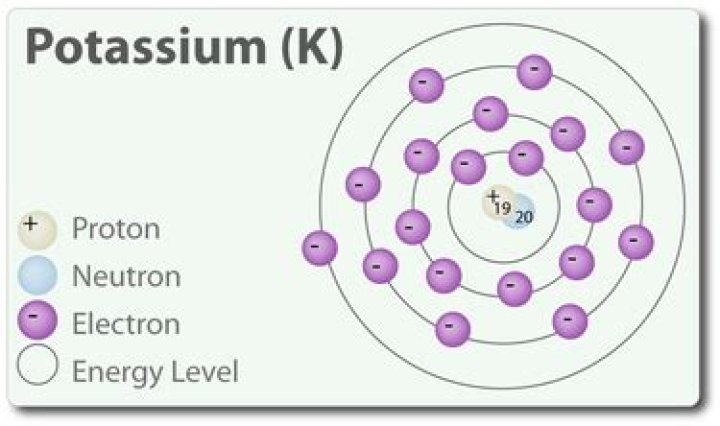
19 protons
20 neutrons
19 electrons
In the ground state, element number = number of protons and electrons. Take out a proton, it has a different element. Get rid of electrons, one element, it just becomes an ion. Number of neutrons = atom m minus atomic number together we get: proton = 19 electrons = 19 neutrons = 20
How Many Neutrons Does Potassium Have
How Many Neutrons Does Potassium Have
How many neutrons, protons, and electrons are a-t-il atium?
Saying the same thing over and over again doesn't help much. But I will not do that.
How Many Neutrons Does Potassium Have
How Many Neutrons Does Potassium Have
IUMK
Series 19
Atom M39.10
19 protons
20 neutrons
19 electrons
Element number = number of protons and electrons in the ground state. Take out a proton, it has a different element. Get rid of electrons, the same element, it just becomes an ion. Number of neutrons = atom m minus atom number together we get: proton = 19 electrons = 19 neutrons = 20
Use Table IC to get the sequence number (19) and the sequence number m (39.1).
Atom number is the number of protons and also the number of electrons in a neutral atom: 19.
Atom MM is a combination of protons and neutrons, 39.1. Subtract the number of protons from atom M, 39 19 = 20, which is the number of neutrons (use integers).
Proton = 19
Neutron = 20
Electron = 19
Atomium M
19 protons and electrons, 20 neutrons
ium serial number
How Many Neutrons Does Potassium Have
How Many Neutrons Does Potassium Have
There are 20 neutrons and 19 protons.
There are 20 neutrons and 19 protons.
(Atomic number = number of protons)
(Number of neutrons = atomic number m atomic number)
Atomic number = 19
Atom m = 39 19 = 20