Cobalt Electron Configuration - How To Discuss
Cobalt Electron Configuration
What is the electron configuration of cobalt (III) (Co3 +)?
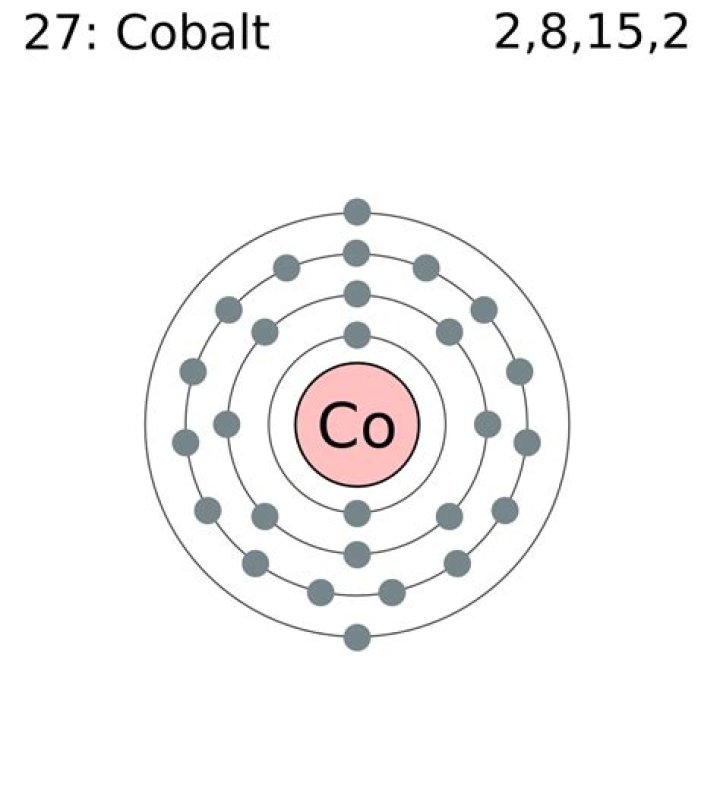
In its ground state, cobalt has the following electron configuration:
[R] 4s2, 3d7.
But when cobalt loses three electrons, it loses 4 and 3. It loses two 3d electrons to make a full 3D, which has a high degree of stability, and loses 14s electrons. 4s and 3d subvelvels have approximately the same amount of energy, so electrons can come out of both surfaces.
The electron configuration for Co 3 + is:
[Ar] 4s1, 3d5.
Co3. Arrangement of electrons
Cobalt Electron Configuration
Cobalt Electron Configuration
What is the electron configuration of cobalt (III) (Co3 +)? 3
I know we should do this normally, but what happens when it's 3+?
In its ground state, cobalt has the following electron configurations:
[Ar] 4s2, 3d7
But when cobalt loses three electrons, it loses 4 and 3. It loses two 3d electrons to make a complete 3d, which has a very high stability, and it loses 14s electrons. The 4s and 3d sub-surfaces have approximately the same energy, so electrons can be released from both sub-surfaces.
The configuration of electrons for Co 3 + is:
[Ar] 4s1, 3d5
Shared electronic configuration
This page can help you.
D:
What is the electron configuration of cobalt (III) (Co3 +)?
I know we should do this normally, but what happens when it's 3+?
Cobalt Electron Configuration
Cobalt Electron Configuration
Co: 4s2 3d7
Co2 +: 4s0 3d7
Co3 +: 4s0 3W6
External 4 is removed by 3d electrons.
The first 2 electrons are missing from the column.
The third electron is missing from orbit.
So 1s2 2s2 2p6 3s2 3p6 3d6
For metals, always remove the first s electron before releasing the D electron. In the case of Cobalt (III), the sequence is as follows:
Co [Ar] 4 s 2 3d 7
Co (III) [Ar] 3d 6
Please answer below.
3d6 4s0 Electrons are lost and then d