Charged atom - How To Discuss
Charged atom
What is an uncharged atom? Uncharged atoms usually contain nuclei of protons and neutrons surrounded by a cloud of electrons. The positive charge of the protons attracts negative electrons and keeps them in orbit.
Are negatively charged atoms?
The atom is electrically neutral and has no general electrical charge. However, each atom contains even smaller particles called electrons. Each electron has a negative charge. When an atom grabs an electron, it becomes negatively charged. When an atom loses an electron, it becomes positively charged.
What is the composition of an atom?
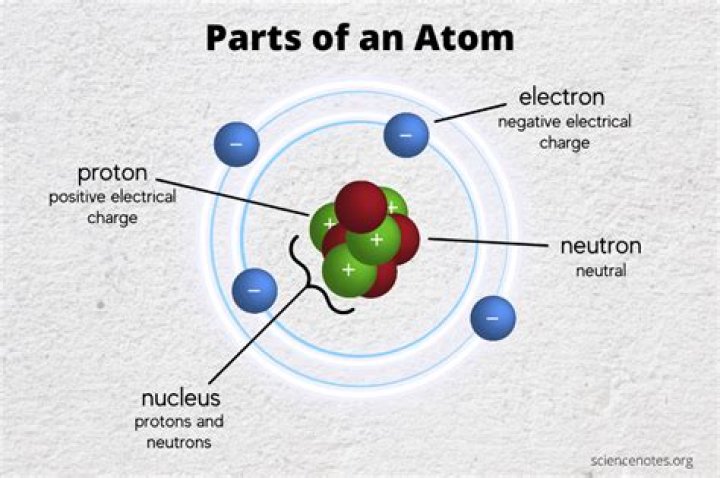
Atoms are made up of three main particles: protons, electrons and neutrons. The nucleus (center) of an atom contains protons (positively charged) and neutrons (uncharged).
What is the name of uncharged particles in an atom?
Atomic Particles Atoms are made up of three main particles: protons, electrons and neutrons. The nucleus (center) of an atom contains protons (positively charged) and neutrons (uncharged). The outer regions of an atom are called electron shells and contain electrons (negatively charged).
Which atom has the greatest nuclear charge?
Therefore, the sodium cation has the highest effective nuclear charge. This also assumes that Na + has the smallest radius of its kind, and for good reason. What is the effective attractive force Z e f f of the valence electrons in the sodium anion, the neutral sodium atom and the sodium cation?
What does atom have no charge?
As mentioned in the introduction to this article, an atom consists mainly of empty space. The nucleus is the positively charged center of an atom and contains most of its mass. It consists of protons, which have a positive charge, and neutrons, which have no charge.
![:brown_circle: :brown_circle:]() What is the uncharged particle in the nucleus of an atom?
What is the uncharged particle in the nucleus of an atom?
A neutron is an uncharged particle in the nucleus of all atoms EXCEPT hydrogen. For a given element, the mass number is the number of protons and neutrons (nucleons) in the nucleus.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is an uncharged atom called
What is an uncharged atom called
Neutrons are uncharged particles that occur in all atomic nuclei (except hydrogen). The mass of the neutron is slightly greater than the mass of the proton. Like protons, neutrons are made up of quarks: one "up" quark (with 2/3 positive charge) and two "down" quarks (each with a third negative negative charge).
What is an uncharged particle found in the nucleus of an atom called?
What is an uncharged particle in an atomic nucleus called? Protons and neutrons accumulate in the nucleus, while electrons revolve around the nucleus. Protons are positively charged particles and electrons are negatively charged particles. Join to unlock this answer! create an account.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Which is the smallest unit of matter with no charge?
Which is the smallest unit of matter with no charge?
It is free. It has the same mass as a proton or weighs 1 amu. An atom is the smallest unit of matter that retains all the chemical properties of an element. Atoms combine to form molecules, which then interact to form solids, gases or liquids.
![:brown_circle: :brown_circle:]() How do electrons contribute to the charge of an atom?
How do electrons contribute to the charge of an atom?
Electrons contribute greatly to the charge of an atom because each electron has a negative charge that corresponds to the positive charge of a proton. Scientists define these charges as "+1" and "1". In an uncharged neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons in the nucleus.
Which is an example of the structure of an atom?
For example, water is made up of hydrogen and oxygen atoms that have combined to form water molecules. Many biological processes break down molecules into individual atoms so that they can be put together into a more useful molecule. Atoms are made up of three main particles: protons, electrons and neutrons.
Does atom have more positive charges than negative charges?
An atom that loses electrons has more positive charges (protons) than negative charges (electrons). It has a positive charge. The atom that accepts electrons has more negative particles than positive ones. It has a negative charge.
How does an atom get positively charged?
When the electrons collide with the atoms in the chamber, they cause some of the electrons to be removed or removed from the atoms. The mass of superheated atoms charged with dissociated electrons turns into plasma. When one or more electrons are removed from an atom, it becomes positively charged.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What atom has a negative charge?
What atom has a negative charge?
The neutral chlorine atom was given a negative charge by taking up an electron. Since it has an unequal number of protons and electrons, it is now an ion. And since negatively charged ions are called anions, it now says:.
Why does an atom have a positively charged nucleus?
Since the nucleus consists only of protons and neutrons, it is positively charged. The electrons revolve around the nucleus, the negative charge of which is neutralized by the charge of the nucleus, stabilizing the atom. Consequently, the positive charge of the nucleus plays an important role in ensuring the stability of the atom.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What are negatively charged atoms called
What are negatively charged atoms called
When an atom has the same number of protons and electrons, the total charge is 0. If you add an extra electron, it becomes negatively charged and is called an anion. More pictures of negatively charged atoms.
Are all atoms positively charged?
Ionic education. All atoms contain components called subatomic particles. Neutrons are neutral particles in an atomic nucleus with positively charged protons. The number of protons determines the identity of an element in an atom, and neutrons help determine whether an atom is an isotope.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is an atom with a positive charge?
What is an atom with a positive charge?
An atom or group of atoms with a positive charge is a cation.
What exactly is a positive or a negative charge?
In ordinary matter, the electrons have a negative charge and the protons in the atomic nucleus have a positive charge. If any part of matter contains more electrons than protons, it is negatively charged, if there are fewer, it has positive charge, and if the number is the same, it is neutral.
What is particle has a negative charge?
Simply put, a particle with a positive surface charge is called a positively charged particle. Likewise, any particle with a negative charge on the surface is called a negatively charged particle. A positively charged atom is called a cation and a negatively charged atom is called an anion.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Negatively charged atoms are called
Negatively charged atoms are called
The atom loses or gains a negative charge. These atoms are then called ions. Positive ion. It occurs when an atom loses an electron (negative charge), it has more protons than electrons. Negative ion. It occurs when an atom acquires an electron (negative charge), it has more electrons than protons.
Negatively charged atoms positively
Each electron has a negative charge. When an atom grabs an electron, it becomes negatively charged. When an atom loses an electron, it becomes positively charged. Electrons can move from one substance to another when objects rub against each other.
![:brown_circle: :brown_circle:]() What is a negatively charged particle?
What is a negatively charged particle?
Negative charge is an electrical property of a particle at the subatomic level. Physically, it supports the positively charged particles by creating an electromagnetic field that exhibits some degree of force and keeps the two particles in a combined and reactive state.
What is a charged atom?
An ion is a charged atom or molecule. It is charged because the number of electrons is not equal to the number of protons in an atom or molecule. An atom can acquire a positive or negative charge depending on whether there are more or fewer electrons in the atom than the number of protons in the atom.
What determines the characteristics of an atom?
An important characteristic of an atom is its atomic number, which is defined as the number of protons. The chemical properties of an atom are determined by its atomic number and are indicated by the symbol Z. The total number of nucleons (protons and neutrons) in an atom is its atomic number.
What is the basic structure of the atom?
The basic structure of an atom consists of a small and relatively massive nucleus containing at least one proton and generally one or more neutrons.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Which atom is the smallest atom?
Which atom is the smallest atom?
If you mean more and less mass (which is a measure of the amount of matter), the smallest is a hydrogen atom with one proton and one electron.
![:brown_circle: :brown_circle:]() What are interesting facts about atoms?
What are interesting facts about atoms?
Fun facts about atoms: Atoms contain a dense nucleus surrounded by a cloud of negatively charged electrons. There are positively charged protons in the nucleus and almost all atomic nuclei (except hydrogen1) contain neutrons with charged neutrons.
![:brown_circle: :brown_circle:]() What is the composition of an atom of steel
What is the composition of an atom of steel
The main component of steel is iron, a metal that is not much ■■■■■■ than copper in its pure form. Except in very extreme cases, iron in the solid state is polycrystalline like all other metals, that is, it is composed of many crystals found at the edges.
![:brown_circle: :brown_circle:]() What are the two main elements of steel?
What are the two main elements of steel?
Carbon steel or normal carbon steel is a metal alloy. It is a combination of two elements: iron and carbon. Other elements are present in amounts too small to affect their properties. Only the following elements are allowed in normal carbon steel: Manganese (max.), Silicon (max.) and Copper (max.).
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What elements are in steel?
What elements are in steel?
The elements that make up stainless steel are nickel, iron, chromium and manganese. The important elements of stainless steel are iron (Fe), carbon (C) and chromium (Cr). Iron, chromium, nickel, carbon. This is a mishmash. Stainless steel is an alloy, a mixture (mixed glass) of metals.
What solid is the main constituent of steel?
The main component of steel is iron, a metal that is not much ■■■■■■ than copper in its pure form. Except in very extreme cases, iron in the solid state is polycrystalline like all other metals, that is, it is composed of many crystals found at the edges.
What is the best steel?
- Series 400. 420 Steel: This steel is common for cheap knives and contains about 0.38% carbon.
- OUT series. The biggest improvement of the AUS series (made in Japan) over the 400 series is the addition of vanadium, which improves wear resistance and offers high toughness.
- ATS series.
- SXXV series.
- Other known steels.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is the composition of an atom of phosphorus with 16 neutrons
What is the composition of an atom of phosphorus with 16 neutrons
So this element has 15 protons (atomic number), 15 electrons and 16 neutrons (mass number minus atomic number). This isotope is phosphorus31, atomic number 15. Click here for the full answer. And how many electrons and neutrons does phosphorus have?
How many electrons does a white phosphorus atom have?
Phosphorus atoms have 15 electrons and 15 protons with 5 valence electrons in the outer shell. Phosphorus is a very reactive element and is therefore never found as a free element on Earth. White phosphorus is highly reactive and unstable.
How big is the atomic mass of phosphorus?
Atomic Mass of Phosphorus The atomic mass of phosphorus is and the atomic mass is the mass of an atom. The atomic mass or relative mass of an isotope refers to the mass of an individual particle and thus refers to some specific isotope of an element.
How many neutrons and protons does a typical neutral atom have?
That is, there are 15 protons in the phosphorus nucleus, fundamentally positively charged massive particles. This sequence number identifies the elemental identity. If there are 15 protons, then the formation of an isotopic mass of 31 requires 16 neutrons, 16 solid particles with neutral charges in the nuclei of the elements.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Positively or negatively charged atom
Positively or negatively charged atom
An atom consists of a positively charged nucleus surrounded by one or more negatively charged particles - electrons. Positive charges are equal to negative charges, so the atom does not have a full charge, it is electrically neutral. What is the charge of an atom? The total charge on an atom is zero.
What is a charged atom
All atoms are neutral because the charges of protons and electrons neutralize each other. Subatomic particles have charges: protons are positive. Neutrons are neutral (no charge).
How do you find the charge of an atom?
If you look at the periodic table of chemical elements in order of atomic number, you will see that the elements on the left are usually positively charged and those on the right are usually negatively charged. To calculate the formal charge of an atom, the formula is used: FC = GN UE 1/2 BE.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How does an atom get a charge?
How does an atom get a charge?
An electric charge arises in an atom of a substance when the number of protons in a nucleus differs from the number of electrons surrounding that nucleus. If there are more electrons than protons, the atom is negatively charged. If there are fewer electrons than protons, the atom has a positive charge.
![:brown_circle: :brown_circle:]() What is an atom that carries a charge called?
What is an atom that carries a charge called?
An atom with a positive or negative charge is called an atomic ion. Atoms of the same element, which have different numbers of neutrons, are called isotopes. A single particle of any element on the periodic table is an atom.
What is the name given to a charged atom?
If an atom has more or fewer electrons than its atomic number, it generally has a negative or positive charge; a charged atom is called an ion. Electrons have been known since the late 1800s, thanks in large part to Thomson; see History of subatomic physics for more details.
![:brown_circle: :brown_circle:]() Which parts of an atom are charged?
Which parts of an atom are charged?
The parts of an atom are protons, electrons and neutrons. The proton is positively charged and is located in the center or nucleus of an atom. All atoms of an element have the same number of protons.
What did Sigmund Freud think about the psyche?
Perhaps Freud's most enduring and important idea was that the human psyche (personality) has more than one aspect. Freud's theory of personality (1923) viewed the psyche as structured in three parts (three parts): Id, Ego, and Super-Ego, all of which develop at different stages of life.
How does Freud explain the three components of personality?
Each component makes its own unique contribution to personality, and these three components work together in ways that have a powerful impact on the individual. Each element of the personality manifests itself at a different time in life. According to Freud's theory, certain aspects of your personality are more important and can motivate you to satisfy your most basic desires.
What did Freud mean by the id, ego and superego?
According to Freud's theory of personality (1923), the psyche is structured in three parts (three parts): Id, Ego, and Super-ego, all of which develop at different stages of life. They are systems, not parts of the brain or physical in any way.
How is the human mind structured according to Freud?
However, he believed that these death instincts were largely mitigated by the life instincts. In Freud's theory, the human mind is divided into two main parts: the conscious mind and the unconscious mind. Consciousness includes all things that a person is aware of or that can be easily realized.
An electrically charged atom
You see, an electrically charged atom is an atom with pure electrical charges, called ions. Positive ions are called cations and unwanted ions are called anions. In fact, atoms have two types of numerical particles:.
What atom has an electric charge?
Parts of an atom are electrically charged. Each proton has a positive electrical charge and each electron has a negative electrical charge. Neutrons are free. In most cases, an atom has the same number of protons and electrons.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is an atom with an electric charge called?
What is an atom with an electric charge called?
An ion (/aɪɒn, ən/) is an atom or molecule with a pure electrical charge. Since the charge of an electron (traditionally considered negative) is equal to and opposite to the charge of a proton (considered positive by convention), the total charge of an ion is not zero because the total number of electrons differs from the number.
What is an atom that carry no electrical charge?
Atoms are made up of particles called protons, electrons and neutrons. Protons carry a positive electric charge, electrons carry a negative electric charge, and neutrons carry no electric charge.
![:brown_circle: :brown_circle:]() What electrical charge does an atom have?
What electrical charge does an atom have?
Electrical charge. Atoms are usually neutral, meaning they have the same number of electrons and protons. In other words, they have no net electrical charge. This is the lowest possible energy level of an atom, or the so-called ground state.
Electrically charged atom
What is an atom or group of atoms with an electrical charge? Ions, any atom or group of atoms that carries one or more positive or negative electrical charges. Positively charged ions are called cations, negatively charged ions are called anions. Does an atom or group of atoms have a positive charge?
Which particles of an atom carry electric charge?
The three particles that make up atoms are protons, neutrons, and electrons. Protons carry a positive electrical charge and form an atomic nucleus. Every atom must have at least one proton.
Is an atom with an electrical charge called an atom?
Atoms with a net electrical charge are called ions. Positive ions are called cations and negative ions are called anions. Atoms have two types of charged particles:.
![:brown_circle: :brown_circle:]() Negatively charged atom
Negatively charged atom
If an atom has the same number of protons and electrons, its total charge is 0. If an atom gains an extra electron, it becomes negatively charged and is called an anion. When you lose an electron, it becomes positively charged and is called a cation. Which ion has the highest charge?