Ch3cl Lewis Structure - How To Discuss
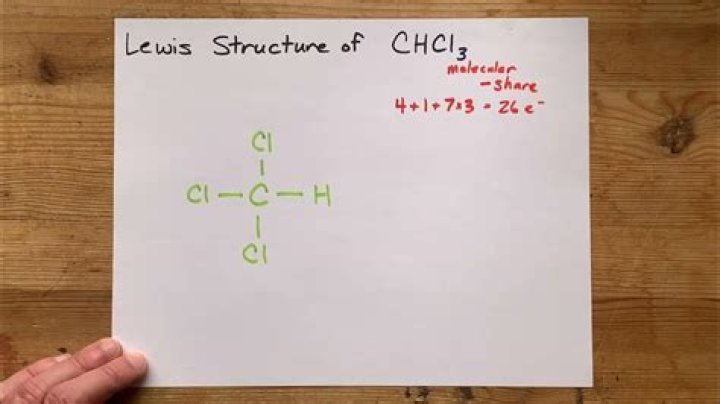
Ch3cl Lewis Structure
Lewis texture for CH3Cl? And would that be a good explanation =]?
A
Step 1: Count the total number of valence electrons
Valence electrons = [Number of atoms] [Number of electrons in the last gas]
  C: [6] [2]  = 4 valence electrons
   H: [1] [0]   = 1 valence electron x 3
   Cl: [17] [10] = 7 valence electrons
Total = 4 + 3 (1) + 7 = 14
Step 2: Infrastructure Design:
The molecule consists of 5 atoms. One of these 5 atoms becomes the central atom and the other 4 are connected to this central atom by the same bond.
But which atom will be the central atom?
H has a valence electron and needs only one more electron to complete its electron, so it can only form one bond. Chlorine has 7 valence electrons and only needs one more electron to complete it, so it can form only one bond. Carbon, on the other hand, has 4 valence electrons and needs 4 more electrons to complete its ■■■■■■■, and can form 4 bonds there. Therefore, carbon will be the central atom:
'Â' Â '' Â 'Â' Â 'Â' Â h
'Â' Â '' Â 'Â' Â 'Â' Â 'Â' |
 C - C ⠀ "H
'Â' Â '' Â 'Â' Â 'Â' 'Â' |
H.
Step 3: Fill in the bytes around each atom
Now that the links have been placed, let's see how many individual pairs we still have. Each bond counts as two electrons, so using 4 bonds, we use 8 of the electrons in steps 1 through 14 to keep the molecule stable.
Acting as a central atom and with four bonds (and up to 2 electrons in each), carbon already has access to 8 electrons around it and can no longer charge. There are also three hydrogens (which can only accept 2 electrons in their 1s). The current chlorine atom has only one relation to carbon, which means that it has access to only 2 ordinary electrons and not 8 stable electrons which they would like. The total atom loses 6 electrons to complete its byte, so we put the remaining 6 valence electrons in the following total.
H.
Â. .Ã'Â '.' Â 'Â'Â' Â |
3: Total - C "" H.
. ™ Â Â Â |
H.
When we count them for the last time, we see that we have used all 14 valence electrons. That's what they're making. That's all there is to it. P help. =)
Lewis structure for Ch3cl
The structure of Ch3cl Lewis
This page can help you.
Come back:
Lewis texture for CH3Cl? And would that be a good explanation =]?
Ch3cl Lewis Structure
Ch3cl Lewis Structure
Lewis's structure for CH3Cl? And an explanation would be nice =]? 3
O
Step 1: Count the total number of valence electrons.
Valence Electron = [Atomic Number] [Number of electrons in the last gas]
  C: [6] [2]  = 4 valence electrons
  H: [1] [0]  = 1 valence electron x 3
L Â CL: [17] [10] = 7 valence electrons
Amount = 4 + 3 (1) + 7 = 14
Step 2: Drag the infrastructure:
The molecule consists of 5 atoms. One of these 5 atoms becomes the central atom and the other 4 are connected to this central atom by single bonds.
But which atom will be the central atom?
H has one valence electron and needs only one more electron to complete its electron, so it can form only one bond. Chlorine has 7 valence electrons and only needs 1 more electron to complete its s, so it can form only one bond. Carbon, on the other hand, has 4 valence electrons and needs 4 more electrons to complete its ■■■■■■■, and can form 4 bonds there. Therefore, carbon will be the central atom:
'Â' Â 'Â' Â 'Â' Â 'H
'Â' '' Â 'Â' Â 'Â' |
 L CL - C - H
'Â' '' Â 'Â' Â 'Â' |
'Â' Â 'Â' Â 'Â' Â 'H
Step 3: Fill in the bytes around each atom.
Now that the links have been placed, let's see how many individual pairs we still have. Each bond counts as two electrons, so using 4 bonds, we use 8 of the 14 electrons in total that we made in step 1 to keep the molecule stable.
Acting as a central atom and surrounded by 4 bonds (and up to 2 electrons in each), carbon already has access to around 8 electrons and can no longer charge. Also complete s in three hydrogen (which can only accept 2 electrons in its 1s). Today's chlorine atom has only one bond with carbon, which means it has access to only 2 ordinary electrons and not to the 8 stable electrons that it would prefer. The Cl atom loses 6 electrons to complete its byte, so we put the remaining 6 valence electrons in Cl as follows:
H.
. .Ã'Â 'Â' 'Â'Â' |
Â: Cl - Câ € "H
™ Â Â |
H.
The last time we counted them, we saw that we used all 14 valence electrons. This is how they are made. That's all you have to do. p help. =)
Lewis framework for Ch3cl
This page can help you.
D:
Lewis's structure for CH3Cl? And an explanation would be nice =]?


