C3h6 Lewis Structure - How To Discuss
C3h6 Lewis Structure
What is the Lewis structure of c3h4?
| Each Lewis scatter plot uses 16 valence electrons and fills the outer shell of each atom. However, atoms can be organized and bonded differently. For the Lewis structure C3H4, calculate the total number of valence electrons for the C3H4 molecule (C3H4 has 16 valence electrons).
What is the shape of c3h4 in this regard?
The bond angle formed by the three carbon atoms is 180 °, which indicates the linear geometry of the central carbon atom. The two terminal carbon atoms are flat and these planes are rotated 90 ° against each other.
Do you also know how many double bonds c3h4 has?
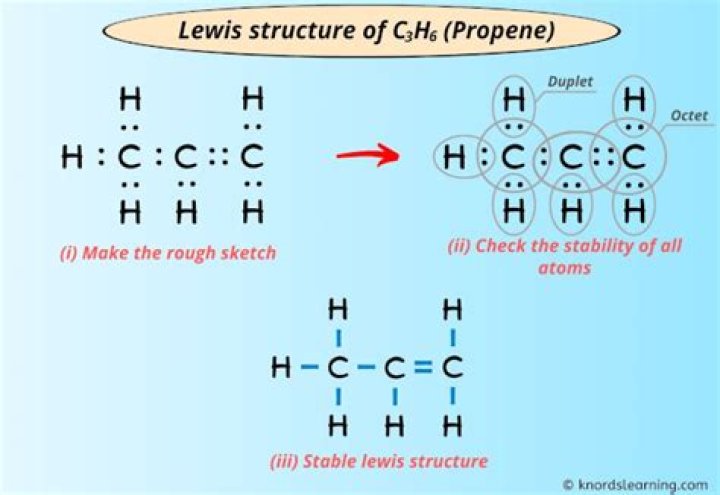
two double bondsSo we can also ask what is the Lewis structure for c3h6?
This is the Lewis C3H6 structure. For C3H6 we have a total of 18 valence electrons. The problem with C3H6 is that there are several ways to draw it based on the chemical formula given here. Let’s take a look at the two ways you can draw the Lewis C3H6 structure.
What is the Lewis structure of c2h4?
Draw the Lewis structure for C2H. For C2H4 you have a total of 12 valence electrons. Designing the Lewis structure for C2H4 (called ethylene) requires the use of a double bond. In a double bond, two pairs of valence electrons share (for a total of four valence electrons).
What is the shape of c4h10?
Tetrahedron
What is the shape of c2h4?
Trigonal plane
What is the hybridization of c3h4?
Alone there are two types of C. The central C is sp hybridized, while the two terminals are sp2 hybridized.
Why is there no c3h4 plan?
Why isn’t the plan alone?
Quora. On the other hand, due to the steric barriers to the overlapping uninhabited porbitals of the double bonded carbon atoms, the carbon chain cannot have parallel hydrogen, as this leads to a higher steric charge.
What are the 5 basic forms of molecules?
Molecular geometries. The VSEPR theory describes five main forms of single molecules: linear, planar trigonal, tetrahedral, bipyramidal trigonal, and octahedral.
How is hybridization determined?
An abbreviation for determining the hybridization of an atom in a molecule How many lone pairs are there in ch4?
If there are all bond pairs, the molecular geometry is tetrahedral (eg CH4). If there is an electron pair and three bond pairs, the resulting molecular geometry is trigonal-pyramidal (eg NH3). With two bond pairs and two lone electron pairs, the molecular geometry is angular or curved (eg H2O).
What is the shape of c3h8?
C3H8 is a linear molecule as it does not have a specific central atom. Since the molecule is linear, it has 180 degree bond angles.
What is the Lewis structure of c2h2?
Draw the Lewis structure for C2H2 (ethylene or acetylene)
What was it called c3h6?
C3H6 may refer to: Compounds that share the empirical formula: cyclopropane. Propene, also called propene.
How do you find the formal accusation?
Formal charge = [# valence electrons on the neutral atom] - [(# pairs of lone electrons) + (½ # bond electrons)] valence electrons = corresponds to the number of groups in the periodic table (for representative elements) . Lonely pairs = single electrons sitting on the atom. Each electron counts as one, so a pair counts as two.
How are Lewis structures drawn?
This guide describes Kelter’s strategy for designing Lewis structures for molecules.
What is propyn hybridization?
There are three carbon atoms in propine, one of which is Sp hybridization and the other two are Sp hybridization. 249 views.
What is the value of hydrogen?
The hydrogen number of hydrogen is one because it has only one valence electron and needs only one distributed electron to fill its energy levels. This means that it can tie into many things. For example, four hydrogen atoms can combine with a carbon atom with four valence electrons to form methane.
What is the dot structure of the Propyn electron?
The molecule shown is propyl. As is known, carbon has 4 valence electrons, hydrogen has 1 valence electron. So the total number of valence electrons in propyne is = 3 (4) +4 (1) = 16. According to the structure of the electron dots, there are 16 bound electrons and 0 unbound electrons. How many resonance structures are there in connection no.
3?
three resonant structures
C3h6 Lewis Structure
C3h6 Lewis Structure
Can you draw the Lewis structure of C3H6? ۔
Mark the hybridization of each carbon atom. Identify all connection angles. Do molecules have many sigma bonds? Is there a PI link? If necessary, circle the pi connection.
(I really only need one sketch, I don't know where it is. Thanks)
CH2 = CH CH3.
The relationship between the first and the second C is 120 ° (sp2)
The ratio between the second and third C is 109.5 ° C (sp3)
This page can help you.
D:
Can you draw the Lewis structure of C3H6?
Mark the hybridization of each carbon atom. Identify all connection angles. Do molecules have many sigma bonds? Is there a PI link? If necessary, circle the pi connection.
(I really only need one sketch, I don't know where it is. Thanks)
C3h6 Lewis Structure
C3h6 Lewis Structure
Can you pull the Lewis structure of C3H6? 3
Mark the hybridization of each carbon atom. Identify all connection angles. How many sigma bonds are there in a molecule? Is there a pi link? If necessary, circle the pi connection.
(I really only need one sketch, I don't know where it is. Thanks)
CH2 = CH CH3
The bond between the first and second C is 120 ° (sp2)
The ratio between the second and third C is 109.5 ° C (sp3)
This page can help you.
D:
Can you pull the Lewis structure of C3H6?
Mark the hybridization of each carbon atom. Identify all connection angles. How many sigma bonds are there in a molecule? Is there a pi link? If necessary, circle the Pi connection.
(I really need a sketch, I don't know where it is. Thanks)
OCCH2CH2CH2COOH is the standard indicator for COOH ic acid, which is actually O = COH, in Y form (bond angle 120).