A 23.6 G Sample Of An Unknown Gas Occupies

A 23.6 G Sample Of An Unknown Gas Occupies
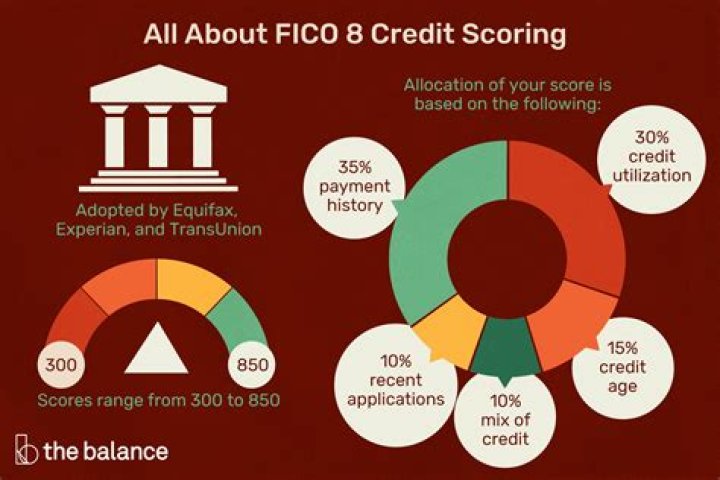
A 23.6 g sample of an unknown gas has a volume of 12.0 liters at standard temperature and pressure. What is this gas molecule? 3
STP condition is 1 atm pressure and 273 K temperature.
PV = nRT
P = 1 atm
V = 12 liters
t =?
R = 0.0821 l atm / K / mol
T = 273 K.
1 x 12 = North x 0.0821 x 273
12 = North X 22,413
n = 12 / 22,413 = 0.535
Number de = quantity of gas / gas m molar = 0.535
23.6 / molar gas m = 0.535
Mرller gas = 23.6 / 0.535 = 44.11 g / mol
The molar is CO2.